Our aim is to verify the association between dietary patterns during early pregnancy and development of gestational diabetes mellitus (GDM) in a low income Brazilian population.
A cohort study followed 841 healthy pregnant women from the 15-20th gestational week until delivery. This study was conducted at the Instituto de Medicina Integral Prof. Fernando Figueira, Brazil. 838 pregnant women during the first half of a healthy pregnancy with a monthly family income below US $ 500.00 were selected. 95 (11.3%) pregnant women developed gestational diabetes mellitus. Three dietary patterns from factor analysis were extracted. The traditional pattern was characterized by dairy products, fruits, vegetable and fish. The mixed pattern included fried food, pizza, juice, manioc flour, red meat and candies. The western pattern was characterized by eggs, white bread, cookies, pasta, pizza, fried food, chicken, candies, chocolate, salty snacks and soft drinks. There were no differences among GDM incidence according to these three dietary patterns. This finding remained after adjustment for maternal age, maternal education, body mass index pre-pregnancy and parity. We concluded that eating patterns studied during early pregnancy were not associated to the development of GDM in this sample of Brazilian pregnant women with low income.
Key words: Dietary patterns; factor analysis; gestational diabetes mellitus; low income population; pregnancy.
Nosso objetivo foi verificar a associação entre o padrão alimentar no começo da gravidez com o desenvolvimento de diabetes mellitus gestacional (DMG) em um grupo populacional de baixa renda no Brasil. Um estudo de coorte seguiu 841 gestantes saudáveis da 15-20 semana gestacional até o parto. Esse estudo foi realizado no Instituto de Medicina Integral Prof. Fernando Figueira (IMIP), Brasil. Oitocentas e trinta e oito gestantes no curso da primeira metade da gestação e com uma renda familiar abaixo de US $ 500.00 foram selecionadas. Noventa e cinco gestantes (11.3%) desenvolveram DMG. Três padrões alimentares distintos foram extraídos através da técnica do fator de análise. O padrão alimentar tradicional foi caracterizado pela ingestão de produtos lácteos, frutas, vegetais e peixes. O padrão alimentar misto incluiu frituras, pizza, sucos, farinha de mandioca, carne vermelha e doces. O padrão alimentar ocidental foi caracterizado por ovos, pão branco, biscoitos, massa, pizza, carne de galinha, doces, chocolate, salgadinhos e refrigerantes. Não foi observado diferença na incidência de DMG entre os três padrões alimentares identificados. Esse achado permaneceu inalterado após o ajuste para a idade e nível de educação maternal, assim como para o IMC antes da gravidez e paridade. Em conclusão, o padrão alimentar materno durante o começo da gravidez entre gestantes de baixa renda não parece estar associado com o desenvolvimento de DMG.
Palavras chave: Padrão alimentar; fator de análise; diabetes gestacional; baixa renda.
Department of Nutrition, Universidade Estadual de Ciências da Saúde de Alagoas (UNCISAL). Biostatistics Department, Mother and Child Health Care, Instituto de Medicina Integral Prof. Fernando Figueira (IMIP), Recife, Brazil.
Gestational diabetes mellitus (GDM) is the most common pregnancy metabolic complication worldwide and its incidence has risen over time (1). Recently the International Association of the Diabetes and Pregnancy Study Groups (IADPSG) made new recommendations for GDM diagnostic with a lower cut-off values in attempt to prevent adverse perinatal outcomes (2). Based on IADPSG criteria the current GDM incidence is above 10% in many countries including Brazil (3). GDM increases the risk for pregnant women to develop type 2 diabetes and hypertensive disorders after delivery besides their offspring having a higher risk of obesity and metabolic disease during adult life (4). For these reasons, the early identification of risk factors for GDM prevention is justified.
Dietary patterns have been associated with the development of several diseases including type 2 diabetes (5). Some studies have assessed the relationship between dietary patterns during early pregnancy and GDM (6, 7). Recently, He et al (8) found that a vegetable pattern was associated with a decreased risk of GDM in China, while sweets and seafood patterns were associated with an increased risk of GDM. However Radesky et al (6) did not find an association between food intake in early pregnancy and the risk of GDM differently of Karamanos et al (7). These findings seem to indicate that the association between dietary patterns and GDM requires further studies.
Dietary preferences are influenced by cultural and environmental determinants (9) and for our best knowledge no study has examined the association between dietary patterns and GDM in South America. In the last decades the typical Brazilian meals which were composed by beans, rice, pasta, roots, tubers, meat (beef or chicken) and eggs, has been changed to ready-to-heat ultra-processed foods (10). Furthermore a low income population is more exposed to unhealthy foods (11) and a large portion of Brazilian population still lives at risk of poverty (12).The aim of this study is to verify the association between dietary patterns during early pregnancy and development of GDM in a low income Brazilian population.
This prospective cohort study followed pregnant women from the second gestation trimester until delivery. This study was conducted at a prenatal health care clinic at the Instituto de Medicina Integral Prof. Fernando Figueira (IMIP), Recife, Brazil. IMIP registers about 6,000 births per year, with all patients from the Brazilian public health system. The enrollment of pregnant women occurred between November 2011 and February 2014.
The sample size calculation was performed using the resource STATCALC the EPI-INFO version 3.5.1. Assuming an incidence of gestational diabetes, 15%, based on the criteria developed by the International Association of the Diabetes and Pregnancy Study Groups ,IADPSG, (13), a statistical significance level of 5% and a type 2 error rate of 20% estimated that it would be necessary to evaluate 717 pregnant women.
Pregnant women were recruited according to the following criteria: an annual income below US $ 6,000.00, a single pregnancy with gestational age between 15 to 20 weeks, aging 18 to 40 years, resident in Recife, and had a low medical risk pregnancy i.e a woman without any previous underlying disease and with prenatal follow-up results within normality parameters. Women who had twin pregnancies (n=11) or a miscarriage (n=7), were diagnosed with a chronic disease (n=27), had a energy intake exceeded 6000Kcal (n=14) or less than 600 (n=8), did not have the oral glucose tolerance test (OGTT) measurement (n=66) were excluded from analyses. The final sample was composed of 841 healthy pregnant women.
All women were measured by a stadiometer and weighed by a digital scale (Filizola Ltd. Brazil) and body mass index (BMI) was calculated [weight (Kg)/stature (m2)]. During the initial interview, study staff completed a structured question to obtain the following variables: maternal age (years), monthly income, education (years of schooling), family history of diabetes mellitus, and parity (number of deliveries).
GDM diagnosis was based on the criteria developed by the IADPSG, after a 75 g, 2 h OGTT for GDM screening at a prenatal care visit at 24 to 28 gestational weeks; fasting >92mg/dl or 1 h >180 mg/dl or 2 h >153mg/dl (13). All GDM women were weekly or monthly accompanied by blood glucose levels, depending on the type of treatment and glycemic control.
The diet was assessed once at enrollment and was considered as representative of one week. A validated food frequency questionnaire (FFQ) was used asking consumption during the previous week, including weekend days, of 81 food and drink items (14 ). The FFQ included food photographs to indicate standard portion sizes: small, medium and large. Interviewers were all health professionals and were previously trained in FFQ application by IMIP nutrition team.
The FFQ answers were converted into grams and total daily energy intake were estimated (Kcal). Nutrient values were obtained from “Programa de Apoio à Nutrição” from Federal University of São Paulo. (http://www2.unifesp.br/dis/produtos/nutwin/). 81 food items were aggregated into 23 food groups based on similarities in the nutritional composition. The 23 food groups are as follow: (i) rice: rice, beans; (ii) pasta: spaghetti, lasagna, gnocchi, ravioli; (iii) pizza; (iv) cookies: cakes, cookies/crackers; (v) white bread; (vi) cassava: cassava, yam, flour, polenta; (vii) coffee: coffee, tea; (viii) red meat: beef, pork, barbecue; (ix) chicken: chicken, giblets, viscera; (x) fish; (xi) vegetables: onion, garlic, pepper, lettuce, cabbage, kale, cauliflower, broccoli; (xii) legumes: tomato, pumpkin, chayote, carrot, cucumber; (xiii) milk: milk, yogurt; (xiv) cheese: cheese curd, yellow cheese, butter; (xv) fruit- banana, orange, tangerine, papaya, watermelon, melon, pineapple, cashew, mango, apple, grapes; (xvi) juice: fruit juices, pulp; (xvii) savory: snacks, popcorn, peanut, cashew nut; (xviii) fried food: French fries, chips, straw potatoes, fried/baked salted pastries; (xix) yogurt; (xx) sweet: sugar, sweet dairy, ice cream, candies, caramels; (xxi) soft drinks; (xxii) canned foods: canned vegetables; (xxiii) chocolate: powdered chocolate, chocolate bars, bonbon, fruit jam, jelly.
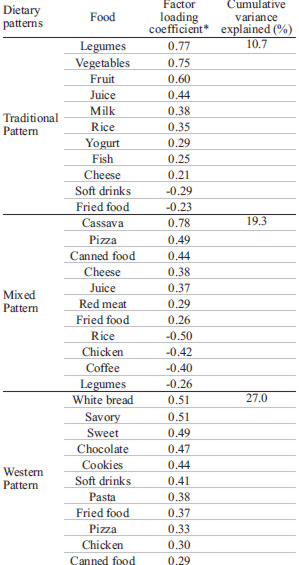
To derive the dietary patterns, we used the principal component factor analysis with varimax orthogonal rotation method (15). We retained three factors, based on Velicer’s Minimum Average Partial (MAP) procedure for determining the number of principal component to extract. These three factors together accounted for 27% of total variation. Food with loadings > 0.20 or < 0.20 on a factor were used to describe the dietary pattern. Factors score were created by the regression method. Due to skewness presence in factor scores, we used Jonckheere-Terpstra test to evaluate mean trend in those factors across ordered categories of maternal age, maternal education and pre-pregnancy BMI; we used the nonparametric permutation test for groups with two categories, gestational diabetes (Yes/No), family history of diabetes (Yes/No) and parity (0/ 1). Poisson regression models were used to examine the associations between dietary pattern scores and the risk of gestational diabetes, adjusting for maternal age, maternal education, pre-pregnancy BMI, family history of diabetes and parity. For all tests, a two-tailed P value < 0.05 was considered statistically significant. All analysis were performed using Stata 12.1SE.
The mean age (SD) of the 841 participants was 26.2 5.8 years. All participants had a monthly family income below US $ 500.00; 90.1% was below US $ 450.00. In addition, 84.8% of the participants had an elementary level schooling, 43.8% had at least one child (range: 1 to 9); 54.9% had a family history of diabetes, 27% were overweight and 14.0% were obese. The incidence of GDM according to the IADPSG was 10.8% (95/841). All GDM diagnostic were performed between 24 and 28 gestational weeks (26.4±0.8) The mean difference in time between dietary pattern scores and OGTT for GDM screening at a prenatal care visit was 8.7±2.2 weeks. Some biological and socio-economic variables of GDM and no-GDM participants are shown at Table 1. GDM women were slightly older.
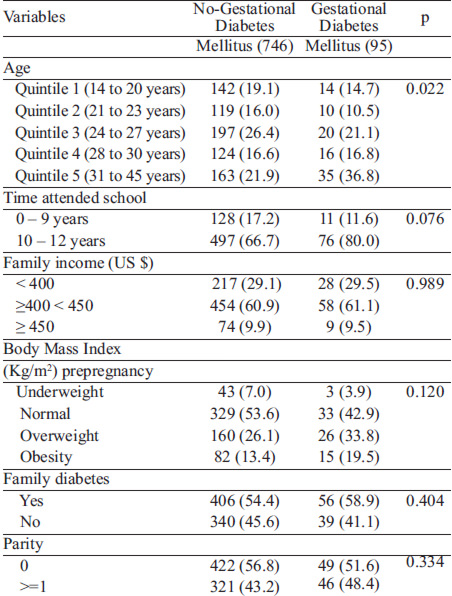
Energy intake was similar among GDM and no-GDM women, respectively 2102 (±794) and 1993 (±742) Kcal. In this study, we extracted three dietary patterns from factor analysis. The factor loadings for each dietary pattern are listed in Table 2. The first pattern (traditional) explained 11.5% of the total variance of food intake. It was characterized by frequent intake of milk, yogurt, juice, fruit, green leaves, vegetable and fish. The second dietary pattern (mixed), which was denoted pattern 2 explained 9.3% of total variance and had positive loadings on cheese, pizza, manioc flour, red meat, and canned food; negative loadings on rice, chicken and coffee. The high-loading foods on the third factor were, eggs, white bread, savory, pasta, fried food, candies, chocolate, salty snacks and soft drinks. The third dietary pattern was denoted western pattern. It explained 8.3% of total variation. The three dietary patterns together accounted for 29.1% of the total variation (Table 2). Factor scores of the three extracted dietary patterns showed no differences between groups with and without GDM (Permutation test p>0.220).
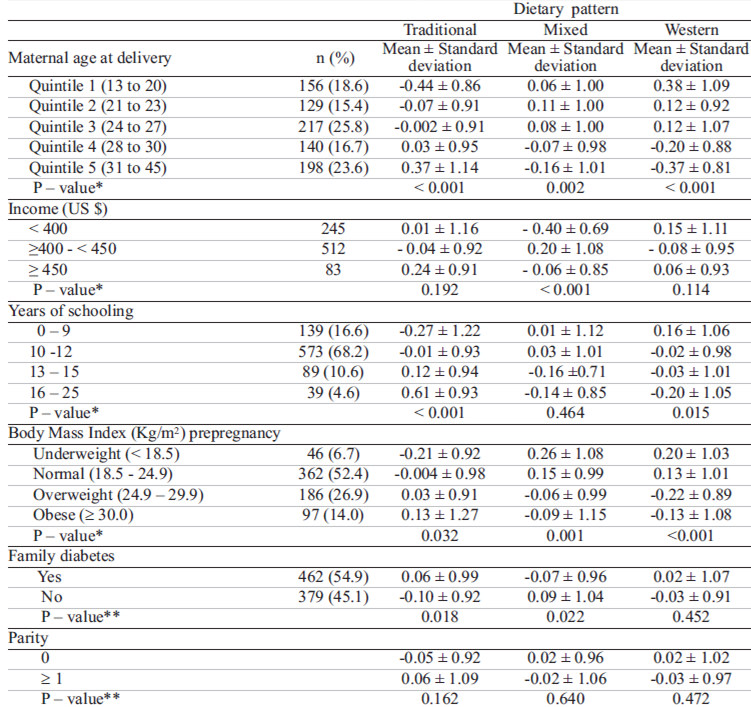
Within each dietary pattern, we verified the distribution of some participant characteristics (Table 4). For the traditional dietary pattern the mean factor scores increased with maternal age at delivery and maternal education. The mean of this factor was higher for those participants that had a history family of diabetes, but there was no association with parity (0/>=1) nor with monthly income. The mixed dietary pattern score was positively associated with monthly income. The mean of this factor was higher for those participants with no history of family diabetes, but there was no association with parity, maternal age at delivery and maternal education. The mean score of the western dietary pattern was inversely associated with maternal age at delivery, income, maternal education and BMI prepregnancy. None of the variables, family history of diabetes and parity were associated with pattern.
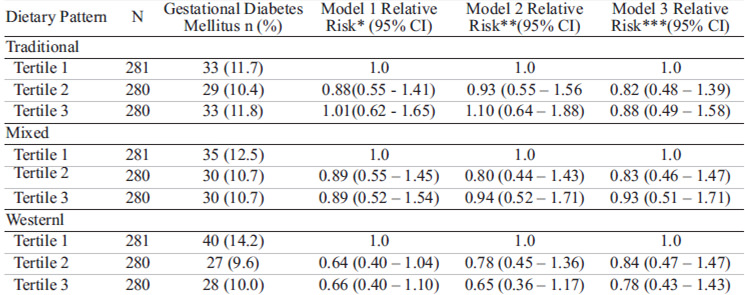
In the univariate analysis there were no significant associations of dietary pattern scores and gestational diabetes. This conclusion remained even after adjustment for maternal age, maternal education, prepregnancy BMI and parity (Table 5).
This study investigated the association between dietary patterns and GDM among low income pregnant women during early pregnancy. To our knowledge this is the first time that this association was studied in a low income population in Latin America. We identified three different dietary patterns, a traditional, a mixed and an western, and none of them were associated with GDM incidence. Our traditional dietary pattern consisted of a consumption of vegetables, fruit, dairy products and rice , while the western pattern represented a consumption of bread, savory, sweets, pasta, fried foods, chicken and canned food . Our mixed dietary pattern included cassava, pizza, canned food, red meat and fried food. Our dietary patterns have some similarities with other eating patterns described elsewhere in the world.
However we studied only women with a low income and dietary patterns have been described as associated with social economic level (12). Population with a higher income and education level are more likely to consume a better quality dietary intake (16 ). In Brazil healthy foods usually have a high cost compared to unhealthy foods. This creates barriers to healthy foods access by Brazilian low-income population. Furthermore dietary patterns have changed in the last decades in Brazil; an increase in the diet's saturated fat and sugar content associated with a decrease consumption of beans, vegetables, fruits and complex carbohydrates (17). In developing countries a high socio economic status or living in urban areas is associated with overall healthier dietary patterns (18). Diet quality follows a socioeconomic gradient highlighting how disadvantaged people present higher rates of obesity and diabetes (11).
Mediterranean Diet, Dietary Approaches to Stop Hypertension (DASH), and alternate Healthy Eating Index, have been associated with type 2 diabetes lower risk (19, 20 , 21). Tobias et al (22) found that prepregnancy adherence to healthful dietary patterns was significantly associated with a lower risk of GDM but this inverse association was partly mediated by BMI. Differently from Radesky et al (6), Zhang et al (23) found that a high intake of processed and red meat was associated with GDM. However according Radesky et al (6), BMI before pregnancy is probably more important than pregnancy diet in development of GDM. Bao et al (24) found that a prepregnancy low-carbohydrate dietary pattern with high protein and animal fat sources is positively associated with GDM risk. A recent systematic review showed that dietary interventions may cause small but significant differences in pregnancy outcomes as a reduction in preterm birth but high-quality randomized clinical trials are required to identify maternal diet intakes that optimise pregnancy outcomes (25). In summary the role of dietary patterns on development of GDM is based on a limited number of studies that are heterogeneous in design, exposure, and outcome measures (26).Dietary patterns obtained using PCA has been debated because this exploratory method lack the repeatability between studies (27). However dietary habits are population-specific and receive strong cultural influences. The differences in study findings might be partly explained by differences in race/ethnicity between study populations.
Our study has some limitations. First a recall bias inherent to the method of FFQ (28) and we assessed food intake during the past week. Second, the diet was assessed only once at enrollment and we could not guarantee that these pregnant women had changed their dietary patterns during pregnancy. However, some studies have shown that overall dietary patterns remain stable during pregnancy (29, 30). Third, pregnant women with higher BMI could under-report food intake. Finally, other healthy lifestyle factors may contribute for positive results in nutritional epidemiology studies, but this is not likely to be a source of confounding in our largely null study.
A prospective cohort design with a large number of pregnant women was conducted though our sample size was calculated to find a prevalence of women with GDM but not in relation to the relationship between groups with different dietary patterns and the development of GDM. Adequate instruments and procedures were used to assess and analyze the dietary intake. Furthermore, to our best knowledge, this is the first study to evaluate the association between gestational dietary patterns and GDM incidence among pregnant women with a low income in Latin America. Dietary habits are population-specific and other studies with this same aim are needed to have a better understanding of the dietary patterns on GDM development. Besides we did not focus on a specific nutrient or a isolated food item but we performed a broader view of food and nutrient consumption through the dietary pattern analysis and examine the effects of overall diet on GDM.
We concluded that in this sample of Brazilian women of low income, there was no association between eating pattern and development of GDM. . This finding seems to provide evidence that specific populations groups may have distinct dietary patterns influencing differently GDM development. Further studies in others populations are needed to clarify this association between dietary patterns and GDM risk.
This work was supported by the Cnpq (grant number 513751–2010–0).
Recibido: 18-05-2016
Aceptado: 22-08-2016