INTRODUCTION
A freshwater fish, Nile tilapia is widely distributed and is one of the most commercially cultured species, being the sixth most cultured finfish species in the world. It is well known for its fast growth, ability to grow in a wide range of culturing conditions, and its high consumer acceptability (1).
Recent studies have indicated that some parts of fish not used as food are appropriate for human nutrition (2) and may be used in oil extraction. Research has shown the existence of significant concentrations of n-3 polyunsaturated fatty acids (n-3 PUFA) in viscera (3), heads (4), and liver (5).
The importance of fish as a source of omega-3 polyunsaturated fatty acids (n-3 PUFA) in human nutrition is widely recognized (6, 7). Among these acids, mainly eicosapentaenoic acid (EPA, 20:5n-3), docosahexaenoic acid (DHA, 22:6n-3), as well as its precursor, alpha linolenic acid (LNA, 18:3n-3) stand out. These acids are associated to numberless benefits to human health. DHA plays an important role in the formation, development, and working of the brain and retina (8). EPA has anti-inflammatory properties (7), and in general contributes to the prevention of heart diseases and to the reduction of biochemical factors associated to cancer (9).
Flaxseed oil is one of the word’s most important vegetable sources of LNA (10), a precursor of the n-3 PUFA series in freshwater fish (11). In a recent study, n-3 PUFA composition of feeds at supply directly reflected in the fatty acid composition in Nile tilapia given flaxseed oil, presenting the highest content of n-3 PUFA in muscle tissue (12).
Considering the few studies related with fatty acid manipulation in fish liver and alternative sources of n-3 PUFA, this work investigated the incremental addition of flaxseed oil in substitution of sunflower oil in feed and its influence on the concentrations of LNA, EPA, and DHA (in mg/g of total lipids) in liver of Nile tilapia (Oreochromis niloticus) maintained in captivity for five months.
MATERIALS AND METHODS
Animal and diets
The experiments were carried out in the Aquaculture Laboratory of the Biology Department of Universidade Estadual de Maringá, Brazil. It utilized 125 Nile tilapia (Oreochromis niloticus) with initial mean individual weights of 88 ± 6 g distributed in 25 ponds (1000-L water capacity) in five treatments and five duplications. The treatments consisted of the addition of flaxseed oil (0%, 1.25%, 2.50%, 3.75%, and 5.00%) in substitution for sunflower oil (control) in feeds (Table 1). After five months, the fish were slaughtered and the liver was removed and kept in polyethylene packing (in N2 atmosphere) at -18 °C. At the beginning of each analysis, the samples were allowed to equilibrate to room temperature and homogenized.
Proximate composition
Proximate composition of liver and feeds were determined as described by Cunniff (13). Total lipids (TL) were determined by Bligh & Dyer (14).
Fatty acid composition
Fatty acid methyl esters (FAME) were prepared by methylation of total lipids by Joseph & Ackman (15). Methyl esters were separated by gas chromatography using a Varian 3300 (USA) gas chromatographer fitted with a flame ionization detector (FID) and a fused-silica DB-WAX capillary column (30 m x 0.25 mm i.d.) (J&W Scientific, Folsom, CA). The operation parameters were as follows: detector temperature, 280oC; injection port temperature, 250oC; column temperature, 170oC for 16 min at 2oC/min up to 210oC with final holding time of 25 min; carrier gas, hydrogen at 0.8 mL/min with linear velocity of 38 cm/s and oxygen filter coupled to the feed line; make-up gas, nitrogen at 30 mL/min; split injection, 1:50 ratio (injection in duplicate). For the identification of fatty acids, fatty acid retention times were compared to those of standard methyl esters (Sigma, St. Louis, MO). Equivalent chain-length values (ECL) were used (16, 17), as well as coupled system gas chromatograph-mass spectrometer Shimadzu QP 5000 and electron impact fragmentation at 70 eV. Retention times and peak area percentages were automatically computed in a Varian 4290 integrator.
Quantification of LNA, EPA and DHA
The concentration of n-3 series fatty acids, LNA, EPA, and DHA in mg g-1 of total lipids, in liver was measured against tricosanoic acid methyl ester (23:0) from Sigma (USA) as an internal standard as described by (15). Theoretical FID (flame ionization detector) correction factor values (15, 18, 19) were used to obtain fatty acid concentration values. The following formula was used to calculate the concentrations:
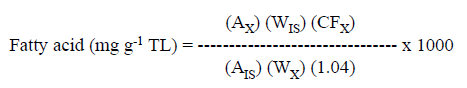
where LT = total lipid, AX is the peak area (LNA, EPA, and DHA), AIS is the peak area of the internal standard (IS) tricosanoic acid methyl ester (23:0), WIS is the weight (mg) of IS added to the sample (in mg), WX is the sample weight (in mg), CFX is the theoretical correction factor, and 1.04 is conversion factor necessary to express results as mg of fatty acids rather than as methyl esters.
Statistics
The values of the means were statistically compared by Tukey test at 5% with one-way ANOVA. Data were processed using the Statistica software (20).
RESULTS
The experimental FID correction factor values for the LNA (0.97 ± 0.02), EPA (0.99 ± 0.01), and DHA (0.98 ± 0.02) were determined experimentally. As the experimental values were close to the calculated ones, the theoretical corrections factors (CFX) of LNA (1.01), EPA (0.99), and DHA (0.97) were used to determine concentrations as recommended by (15, 18).
According (Table 1), there were no significant differences (P > 0.05) in either total lipids or moisture contents of feeds among the treatments. The increase in the concentration of LNA acid was well established; values ranged between 13.6 and 272.4 mg/100 g of feed with a significant difference (P < 0.05) between treatments with flaxseed oil. EPA and DHA were not detected in feeds. Fatty acids profiles of commercial feeds used in treatment of cultured species in Brazil presented low values of LNA (3.3%) and high values of LA -18:2n-6 (38.8%) (21).
TABLE 1
Composition of experimental feeds
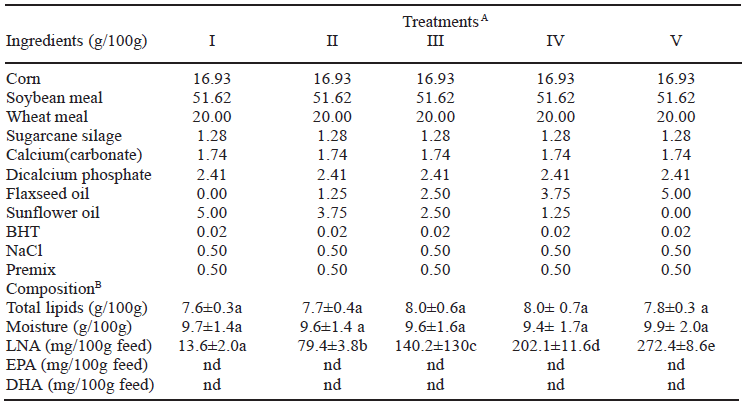
ATreatments: I (0.00%); II (1.25%); III (2.50%); IV (3.75%) and V (5.00%) of flaxseed oil completed up to 5.00% with sunflower oil. BData are presented as mean (n=9) ± SD. Different letters in the same line are significantly different (P<0.05) by Tukey test. Abbreviations: LNA = alpha linolenic acid. EPA = eicosapentaenoic acid. DHA = docosahexaenoic acid. BHT = butylated hydroxytoluene. Premix = mineral and vitamin supplement. nd = not detected.
The liver of Nile tilapia (Table 2) did not present any significant difference between treatments I, III, IV, and V for total lipids and protein contents. However, treatment II (1.25% flaxseed oil) presented the largest lipid content (7.2%) and the lowest protein content (12.1%), differing significantly from other treatments. The values of total lipids and protein in this experiment were lower than those found by researchers (22), who studied the liver of juvenile Nile tilapia, with mean values of 10.2% (total lipids) and 14.3% (protein), while total lipid content was higher than those found in the liver of Atlantic salmon (23). Moisture and ash contents did not vary significantly (P > 0.05) between treatments, with mean values of 72.5% (moisture) and 1.1% (ash). These values are close to those determined by (5) in Nile tilapia, moisture (72.7%) and ash (1.0%).
TABLE 2
Proximate composition (g/100g) and LNA, EPA, and DHA concentrations (mg g-1LT) in liver
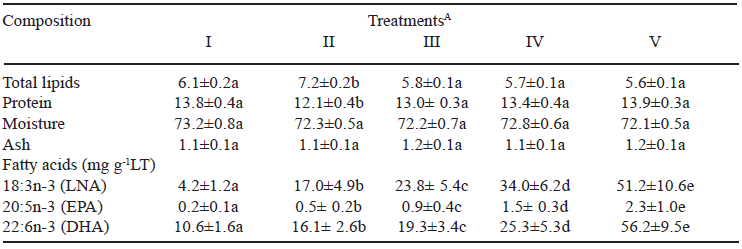
ATreatments: I (0.00%); II (1.25%); III (2.50%); IV (3.75%) and V (5.00%) of flaxseed oil completed up to 5.00% with sunflower oil. Data are presented as mean (n = 30) ± SD. Different letters in the same line are significantly different (P < 0.05) by Tukey test. Abbreviations: LT = total lipids. LNA = alpha linolenic acid. EPA = eicosapentaenoic acid. DHA = docosahexaenoic acid.
DISCUSSION
It was observed differences in the protein concentration and the total lipids between treatments I and II, with an increase in the lipid concentration and a decrease in the protein concentration. After these treatments, the concentrations were stabilized.Total lipids (Table 2) average of 7.8% was larger than 1.1% in fillets (24) and protein average of 13.2% was smaller than 18.0% (25) in fillets of Nile tilapia.
The fatty acid LNA is a precursor of the n-3 PUFA series and only LNA was present and in increasing amounts in the different feeds of this experiment (Table 1). In the elongation and desaturation conversions of the series, EPA and DHA in the liver of Nile tilapia (Table 2) and some LNA were rather stored in the liver rather than converted. Similar results were obtained in muscular tissue (26) and visceras (27) of Nile tilapias.
Increases in the concentrations of LNA, EPA, and DHA (in mg g-1 total lipids) were well established in liver, with a significant difference (P<0.05) between all the treatments with the incremental substitution of sunflower oil by flaxseed oil. Therefore, increasing the amounts of LNA in feed can markedly increase the amounts of EPA and DHA in Nile tilapia liver.
CONCLUSION
There was no significant alteration in the proximate composition, except in treatments I and II. The increase in the concentration of alpha-linolenic acid (LNA) in feeds resulted in a gradual increase in fatty acids alpha-linolenic (LNA), eicosapentaenoic (EPA), and docosahexaenoic (DHA) in all treatments. The largest concentration of these fatty acids in Nile tilapia liver was obtained with treatment V (largest level of flaxseed oil).
REFERENCES
- Yasmin A, Takeuchi T, Hayashi M, Hirota T, Ishizuka W, Ishida S. Effect of conjugated linolenic and docosahexaenoic acids on growth of juvenile tilapia Oreochromis niloticus . Fish Sci 2004; 70: 473-481.
- Pessatti ML. Aproveitamento de subprodutos do pescado. Meta 11. Relatório final de ações prioritárias ao desenvolvimento de pesca e aqüicultura no Sul do Brasil, Convênio Ministério da agricultura, pecuária e abastecimento (MAPA), Universidade do Vale do Itajaí, 2001.
- Souza NE, Matsushita M, Franco MRB, Prado IN, Visentainer JV. Composition química, perfil de ácidos graxos e quantificação dos ácidos alpha-linolênico, eicosapentaenóico e docosahexaenóico em vísceras de tilápias (Oreochromis niloticus). Acta Sci Technol 2005; 27: 73-76.
- Moreira AB, Souza NE, Visentainer JV, Matsushita M. Composição de ácidos graxos e teor de lipídios em cabeças de peixes: Matrinxã (B. Cephalus), Piraputanga (B. Microlepis) e Pracanjuba (B. Orbignyanus) criados em diferentes ambientes. Ciênc Tecnol Aliment 2003; 23: 179-183.
- Carapelli R, Matsushita M, Souza NE, Visentainer JV. Fígados de tilápia: composição centesimal e de ácidos graxos. Rev. Nac Carne 2004; 4: 160-161.
- Simopoulos AP, Leaf A, Salem N. Essentiality andrecommended dietary intakes for omega-6 and omega-3 fatty acids. Ann. Nutr Metabol 1999; 43: 127-130.
- Simopoulos AP. Omega-6/omega-3 essential fatty acid ratio and chronic diseases. Food Rev Int 2004; 20: 77-90.
- Sangiovanni JP, Chew EY. The role of omega-3 long chain polyunsaturated fatty acids in heath and disease of the retina. Progr Retin Eye Res 2005; 24: 87-138.
- Kimura Y, Takaku T, Nakajima S, Okuda H. Effects of carp and tuna oils on 5-fluorouracil-induced antitumor activity and side effects in sarcoma 180-bearing mice. Lipids 2001; 36: 353-359.
- Wanasundara JPD, Shahidi F. Alkanol-amonia-water/hexane extraction of flaxseed. Food Chem 1994; 49: 39-44.
- Hendersom RJ, Tocher DR. Fatty acid metabolism in freshwater fish with particular reference to polyunsaturated fatty acids. Arch. Anim. Nutr 1996; 49: 5-22.
- Justi KC, Hayashi C, Visentainer JV, Souza NE, Matsushita M. The influence of feed supply time on the fatty acid profile of Nile tilapia (Oreochromis niloticus) fed on a diet enriched with n-3 fatty acids. Food Chem 2003; 80: 480-493.
- Cunniff PA. (Ed.). Official Methods of Analysis of AOAC International. 16th ed. Arlington: Association of Official Analytical Chemists. 1998.
- Bligh FG, Dyer WJ. A rapid method of total lipid extraction and purification. Can. J. Bioch and Physiol 1959; 37: 911-917.
- Joseph JD, Ackman RG. Capillary column gas chromatography method for analysis of encapsulated fish oil and fish oil ethyl esters: collaborative study. J AOAC Intern 1992; 75: 488-506.
- Thompson RH. A simplified fatty acid analyses in multicomponent foods with a standard set of isothermal GLC conditions couplet with ECL determinations. J. Chrom Sci 1996; 34: 495-504.
- Strànsky K, Jursík T, Vitek A. Standard equivalent chain length values of monoenic and polyenic (methylene interrupted) fatty acids. J. High Res Chrom 1997; 20: 143-158.
- Bannon CD, Craske JD, Hilliker AE. Analysis of fatty acid methyl ester with high accuracy and reliability. Validation of theoretical relative response factors of unsaturated ester in the flame ionization detector. J. Am Oil Chem Soc 1986; 63: 105-110.
- Visentainer JV, Franco MRB. Ácidos Graxos em óleos e gorduras: Identificação e Quantificação, 1 ed. Varela: São Paulo, Brasil, 2006.
- Statsoft. Statistica 5.1 Software. Tucksa: Statsoft. 1996.
- Moreira AB, Souza NE, Visentainer JV, Matsushita M. Fatty acids profile and cholesterol of three Brazilian brycon freswater fish. J Food Comp Anal 2001; 14: 565-574.
- Visentainer JV, Gomes STM, Silva ABM, Santos-Junior OO, Justi KC, Souza NE, Hayashi C, Matsushita M. Composição físico-química e perfil de ácidos graxos em fígados de tilápias (Oreochromis niloticus) submetidas ao fornecimento de rações diferenciadas à base de óleos de girassol e linhaça. Ann Ass Bras Quim 2003; 51: 18-21.
- Jordal AEO, Lie O. Torstensen BE. Complete replacement of dietary fish oil with a vegetable oil blend affect liverlipid and plasma lipoprotein levels in Atlantic salmon (Salmo salar L.). Aquaculture Nutr 2007; 13: 114-130.
- Visentainer, JV, Souza, NE, Matsushita, M, Hayashi, C, Franco, MRB. Infuence of diets enriched with flaxseed oil on the alfalinolenic (LNA), eicospentaenoic (EPA) and docosahexaenoic (DHA) fatty acids in Nile tilapia (Oreochromis niloticus). Food Chem 2005, 90:557-560.
- Justi, KC, Padre, RG, Hayashi, C, Soares, CM, Visentainer, JV, Souza, NE, Matsushita, M. Efeito da temperatura da água sobre desempenho e perfil de ácidos graxos de tilapia do Nilo (Oreochomis niloticus. Acta Scientiarium 2005, 24:529-534.
- Souza, ES, Matsushita, M, Oliveira, CC, Franco, MRB, Visentainer, JV. Manipulation of fatty acid composition Nile tilapia (Oreochromis niloticus) fillets with flaxseed oil. J Sci Food Agri 2007, 87:1677-1681.
- Visentainer, JV, Matsushita, M, Souza, NE, Prado, IN, Franco, MRB. Composiçao química de ácidos graxos e quantificação dos ácidos alfa-linolenico, eicosapentaenoico e docosahexaenoico em vísceras de tilapias (Oreochromis niloticus. Acta Scientiarium 2005, 27:73-76.