Introduction
Mayonnaise is a semisolid sauce formed by mixing vegetable oil, egg yolk, vinegar and salt and other optional seasonings (1). In the past 100 years it has been considered the most used sauce in the world (2). Its structure, creaminess, appearance, rheological behavior and stability parameters are of great importance for its sensory properties and texture, and hence consumers’ choice and satisfaction (3).
From a colloidal point of view, mayonnaise is an oil-in-water emulsion with low pH, characterized by having very high oil content, ranging from 65 to 85%, depending on the formulation (3).
Because of its acid taste, conventional manufactured mayonnaise has started to be rejected by some consumers and is being replaced by home-made mayonnaise, prepared with raw eggs and generally mixed with boiled potatoes and other vegetables, also known as low-acid mayonnaise (LAM) (4). However, the low acidity, consumption of this mayonnaise can pose a risk of microbial contamination. Nevertheless, many people choose to consume it because of its sensory characteristics (5).
The vegetable oil in the formulation of lowacid mayonnaise has nutritional and economic importance. Its unsaturated and saturated fatty acid composition will determine the oxidative stability of the final product. Soybean oil, for example, is more susceptible to oxidation than many other vegetable oils due to its high linoleic acid content, so the presence of antioxidants is necessary to ensure its oxidative stability (6).
There are several factors that can accelerate the lipid oxidation process of low-acid mayonnaise: the presence of transition metals (even in small amounts), which can accelerate oxidation, reducing the oil induction period and making it more susceptible to oxidation; storage temperature (higher temperatures accelerate the oxidation process); exposure to light, which can cause photolytic self-oxidation; pH; composition (oil concentration); and the use or not of antioxidants (1).
In all segments of the food industry, conservation of food is extremely important and mainly involves controlling the multiplication of microorganisms, which are responsible for the generation of risks to health and food spoilage. Microbial contamination has a significant influence on the quality of food. It can compromise safety due to the presence of pathogenic bacteria as well as conservation status by the multiplication of spoilage bacteria that reduce shelf life (7). Mayonnaise is particularly subject to microbial contamination by Salmonella spp.
There have been several reports in the international literature of health problems caused by homemade mayonnaise produced with raw egg (8). The composition of the mayonnaise and the pH are the factors that most influence its microbiological safety. Therefore, the industry has been using additional barriers to ensure the microbiological quality of mayonnaise as well as its stability. However, the additives used as “barriers” are not pleasing to many consumers, who prefer foods without artificial chemicals, generating a strong demand for products with fewer additives as well as natural substitutes (9).
Mayonnaise consumers are increasingly concerned about synthetic or artificial additives, so “healthier” versions have been developed (10). Butyl hydroxyanisole (BHA) and butylated hydroxytoluene (BHT), are the most commonly used synthetic antioxidants, and show high efficacy, but their use in food has been partially restricted due to their adverse effect on the enzymes in human organs. Accordingly, there is currently great interest throughout the world in finding new antimicrobials and antioxidants from natural sources (11). The main alternatives that have been studied include plant extracts (12). Since the middle ages, essential oils have been widely used in medicinal and cosmetic applications due to their bactericidal, virucidal, fungicidal and insecticidal activities. With the growing preference of consumers for“natural” products, interest in these oils in the pharmaceutical, agricultural and food sectors is growing (13).
Essential oils (EO) are liquid mixtures of volatile compounds derived from aromatic plants, usually by steam distillation. They constitute what is called the “essence” of plants and usually have pleasant fragrances. Essential oils have been used for millennia because of their perceived health benefits and beneficial cosmetic effects, well documented in ancient literature. Some of the beneficial properties described are antiseptic, antioxidant, antimicrobial and anti-inflammatory (14).
Essential oils are complex mixtures of individual compounds. Each of these compounds contributes to the beneficial and adverse effects of these oils. Therefore, knowledge of the essential oil composition allows better applications (15).
Regarding antimicrobial and antioxidant properties, several studies have been performed using different bacteria and fungi. The chemical activity is caused by the presence of terpenes and their oxygenated compounds. Each compound contributes to the biological activity of the essential oil (16). The antimicrobial activity of phenolic compounds present in essential oils increases with acid pH, which makes it essential to find oils suitable for use in low-acid mayonnaise (17,18).
Thus, it is relevant to investigate the possibility of replacing synthetic antioxidants and antimicrobials with essential oils in formulating mayonnaise.
This article focuses on knowledge of the physico-chemical parameters of low-acid mayonnaise, showing the influence of these parameters on the start of lipid oxidation, and the effect of essential oils on lipid oxidation and microbial growth, as well as the causes of deterioration if low-acid mayonnaise, by means of a review of the literature in the SciELO and ScienceDirect databases.
Lipid oxidation
Oxidation of unsaturated fatty acids has been the main focus of research into the instability of emulsions. Like all high-fat foods, low-acid mayonnaise is susceptible to deterioration due to self-oxidation of unsaturated fats in the oil (1).
Lipid oxidation is responsible for the development of unpleasant tastes and odors, making improper for consumption. It also causes other changes that affect not only the nutritional quality due to degradation of fat-soluble vitamins and essential fatty acids, but also integrity and food safety, through the formation of potentially toxic polymeric compounds (18).
Autoxidation is the main mechanism of oxidation of oils and fats. There is a sequence of interrelated reactions to explain the autoxidation process of lipids (19). This autoxidation is associated with the reaction of oxygen with unsaturated fatty acids and occurs in three stages: initiation, propagation and termination. During initiation, the formation of free radicals of fatty acid occurs due to the withdrawal of an allyl carbon hydrogen in the fatty acid molecule in favorable conditions of light and heat. In propagation, free radicals, which are readily susceptible to atmospheric oxygen attack, are converted into other radicals, producing the primary oxidation products (peroxides and hydroperoxides), whose structure depends on the nature of the fatty acids present. The free radicals formed act as reaction propagators, resulting in an autocatalytic process. At the end, the two radicals combine with the formation of stable products (secondary oxidation products) obtained by scission and rearrangement of peroxides (volatile and non-volatile epoxides) (18).
This lowers the nutritional value of food products as well as changes the color, texture and other sensory and physiological properties. The resulting lipid peroxidation from the reaction between the unsaturated fatty acids and molecular oxygen is a serious problem for the oil and fat industry. It not only deteriorates the quality of fatty foods and fatty acids, causing chemical damage, but also produces free and reactive oxygen radicals that are associated with carcinogenesis, mutagenesis, inflammation, aging and cardiovascular disease. Because of this, consumers do not accept oxidized foods, causing losses to food producers (20)
For the purpose of inhibiting or retarding lipid oxidation of oils, fats and fatty foods, chemical compounds known as antioxidants are employed. However, although hundreds of compounds have been proposed to inhibit oxidative deterioration of oxidizable substances, only some can be used in products for human consumption (18).
Factors that influence lipid oxidation and microbial growth
Mayonnaise composition
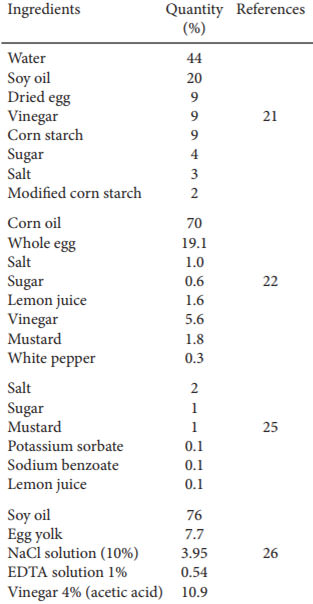
Since 1756, various mayonnaise formulations have been described. Although the basic ingredients have always been the same, many other lesser ingredients can be used (2).
Mayonnaise can be divided according to the function of its components into three phases: oil phase, spice phase and egg yolk phase. The oil phase comprises the vegetable oil used in mayonnaise preparation. Among the most common oils used to make mayonnaise are soybean oil and sunflower oil. The spice phase consists of various seasonings, together with water-soluble food additives and water. The egg yolk phase acts as the emulsifier (21). Table 1 summarizes the various mayonnaise compositions found in the literature. The preparation is carried out with the mixture of egg, vinegar and then all other ingredients except the oil in a mixer or blender until they are homogeneous. Then the oil is gradually added until the sauce begins to thicken (22). Some formulations include ingredients such as mustard, pepper and corn starch. Brazilian law allows other ingredients to be added, provided there is no adulteration of the product (23). U.S. law (21 CFR §169,140) allows the use of spices and monosodium glutamate as long as mayonnaise color is not modified (24).
TABLA 1. Mayonnaise composition
The use of vinegar in mayonnaise preparation is recommended since, in the pH range observed in this food, acetic acid presents a greater number of molecules than coupled citric acid. Furthermore, the addition is suggested of mustard in a concentration from 0.30 to 1.50%, along with garlic, due to the antimicrobial effect of the allyl isothiocyanate and allicin present in these spices, respectively. Vegetables such as carrots can also be added, resulting in absorption of acetic acid by the plant and, consequently, reduced bactericidal effect (27).
Egg yolk is the most problematic ingredient of mayonnaise formulation, since eggs and egg-based products are often associated with outbreaks of food poisoning by Salmonella. The foods most often involved are mayonnaise, ice cream and other cold desserts that are prepared and consumed after addition of raw egg (28)
The yolks have a more favorable environment for bacteria than the whites due to pH and lipid content. Fresh egg yolk has pH values around 6.0, varying very little even in prolonged storage conditions. The physical and chemical changes in the viscosity of the white and permeability of the vitelline membrane is exacerbated by aging. Increased permeability allows Salmonella to reach the yolk. The penetration through the yolk vitelline membrane in experimentally infected eggs can occur after 24 hours at 25 °C. Once in the yolk, Salmonella can multiply at temperatures between 10 to 25 °C, and the number of bacteria increases rapidly at 25 °C (27).
Vertical transmission and horizontal transmission are possible routes by which Salmonella spp. can contaminate intact eggs. Several authors have reported that in birds, the serotype enteritidis can be transmitted vertically. Contamination of eggs by vertical transmission occurs when Salmonella Enteritidis bacteria enter the eggshell during or after oviposition, and contaminate the internal contents (27). It is difficult to completely avoid contact between the egg shell and chicken feces. The extent of fecal contamination of the shell determines the contamination level. Externally contaminated eggs have a risk of internal contamination through horizontal transmission and can cause crosscontamination of other foods in the kitchen (28).
Vinegar is one of the ingredients most often used to form an antimicrobial barrier. It is the most common acid used in the preservation of mayonnaise because it has antiseptic value and also helps prevent deterioration and rancidity (21). Vinegar is usually added together with other acids such as lactic acid to keep the pH of the mayonnaise low (between 3.3 and 3.8). The use of other acids avoids an overly strong acid taste caused by vinegar (29).
Salt improves the taste of the mayonnaise and acts as a preservative. Because salt is dissolved only in the aqueous phase, which is much smaller than the oil phase, it ends up having high concentration, and thus hinders microbial growth (30).
Water activity (Aw)
One of the main components of most foods is water. Water activity (Aw) is a parameter that measures the amount of free water in the food, being defined as the ratio between the partial water vapor pressure contained in the solution or in the food and the standard-state partial vapor pressure of water at a given temperature (31). Just like water activity, the moisture content of a food is very important because it is related to its stability, quality and composition, and can be affected during storage (32). Foods with excessive moisture or water activity near 1 are subject to rapid deterioration. This is the case of low-acid mayonnaise, which has water activity ≈ 0.97, where the emulsion formed is not sufficient to ensure low water activity, making this an essential parameter to determine the survival and growth of pathogens. Control of water activity is one of the oldest techniques of food preservation (33).
The temperature x Aw binomial is also extremely important to control microbial growth, since the exposure of S. Enteritidis to water activity of 0.95 at 21 °C increases the thermal resistance of this bacterium (27).
Hydrogen potential (pH) and acidity
The pH of low-acid mayonnaise varies from 3.6 to 4.0. The best viscoelasticity and stability are observed when the pH is near the isoelectric point of the egg yolk (34). Monitoring pH is essential to ensure the quality of mayonnaise, since a decrease in pH can have pro-oxidant effect, breaking the existing bonds between the proteins of the egg yolk and iron. Subsequently, the iron becomes more accessible to oxidation. Also, the distribution of secondary oxidation products is pH-dependent (35). Low-acid mayonnaise, despite a better taste, degrades quickly at low pH (21). The main risk of low pH is multiplication of Salmonella, since the yolk is a natural culture medium, facilitating survival of the pathogen.
In the refining process, the acidity of vegetable oils is reduced as a quality control measure. With the occurrence of the non-enzymatic oxidation, oil acidity increases. The decrease in mayonnaise pH (increased acidity) during storage can be attributed mainly to the activity of microorganisms that are tolerant to acids, such as the lactic acid bacteria that are present in the aqueous phase in mayonnaise. In addition, these increases can be also be caused by the activity of hydrolytic and oxidative enzymes present in eggs (2).
Essencial oils
Antioxidant activity of essential oils
Spices and herbs are excellent sources of antioxidants and have a long history of use. More than 5,000 years ago, the ancient Egyptians used spices and herbs (cumin, cinnamon and onions, among others) in their food, for medicinal purposes and for mummification. Several studies have shown that spices and herbs such as rosemary, sage and oregano, with their high content of phenolic compounds, serve as strong antioxidants (36).
Antioxidants are compounds that inhibit or delay the onset of oxidation and can be classified as natural or synthetic. Due to their components, spices and herbs are excellent sources of antioxidants for food preservation (36).
Due to market requirements, the use of synthetic antioxidants is being replaced more and more by natural antioxidants from plant sources. Many sources of antioxidants of vegetable origin have been studied in recent years, and numerous types of plants have been identified with various antioxidant activities (37).
It has been clearly demonstrated that plantderived phenolic compounds have antioxidant properties. Research has demonstrated the strong phenolic character of essential oils from oregano (Origanum vulgare), thyme (Thymus vulgaris L.), wild thyme (Thymus serpyllum L.), rosemary (Rosmarinus officinalis), sage (Salvia officinalis), nutmeg (Myristica fragrans), cinnamon (Cinnamomum verum), clove (Syzygium aromaticum), allspice (Pimenta dioica), ginger (Zingiber officinale), turmeric (Curcuma longa) and paprika (Capsicum annuum) (10,37).
Even essential oils with low antioxidant activity can serve as intermediates in forming other compounds with antioxidant activity, such as linalool, which can be obtained from the essential oil of ho-sho. Linalool can be employed as an intermediate in synthesis of vitamin E, which has high antioxidant activity (38).
The antioxidant mechanism of phenolic compounds in lipids has not yet been fully explained (37).
Rosmarinic acid, caffeic acid, coumaric acid, quercetin, thymol and carvacrol are some of the compounds responsible for antioxidant activity of various essential oils (19). Laguori and Boskou (39) concluded that inhibition of oxidation of essential oils of oregano species is highly dependent on carvacrol and thymol content.
Table 2 presents some studies of antioxidant activity in the DPPH test, showing the IC50 (which is the concentration of essential oil required to reach 50% antioxidant activity) of some plants of interest. The larger the IC50, the lower the antioxidant activity of the oil is. In the DPPH test, the essential oil’s ability to act as a donor of hydrogen atoms or electrons in the transformation of DPPH in the reduced form of DPPH - H (diphenyl picryl hydrazine) is measured spectrophotometrically. The DPPH is a free radical stable at room temperature, producing a violet solution in ethanol. In the presence of antioxidant compounds, the DPPH is reduced, producing a clear ethanol solution.
Table 2. Main essential oils with antioxidant activity properties

Table 3 summarizes the antioxidant compounds isolated from herbs and spices and their mode of action to inhibit or delay the oxidation of fats and oils in foods. Some antioxidants derived from spices and herbs react with the free radicals created during the early autoxidation stage. Others form complexes with metal ions (36).
Table 3. Antioxidants isolated from herbs and spices (36)

Antimicrobial activity of essential oils
The antimicrobial effects of essential oils have been investigated against a wide range of microorganisms over the years, but the mechanism of action is still not completely understood (44). Essential oils typically contain many bioactive molecules and can consist of up to 45 different compounds. This structural diversity allows the presence of different modes of action that are responsible for acting in different cellular targets. Essential oils have various modes of action that can result in cell death, including cell wall disorders of bacteria by forming pores that result in increased permeability of the membrane and allow the release of cell components, reduction of intracellular pH and changes in the intracellular concentration of adenosine triphosphate (ATP) (7, 44). The presence of oxygenated monoterpenes, monoterpene hydrocarbons and aldehyde essential oils has the ability to inhibit the process of breathing and movement of ions and consequently leads to destruction of the bacterial cell (45).
The antimicrobial activity of an essential oil is due to the interaction effect between the major and minor compounds of the oil (15). Therefore, one of the most important factors in the study of their applicability is the chemical composition, which can vary within the same plant due to biological factors (genetics, nutrition and development stage) and edaphoclimatic factors (local climate and soil type) (46).
Antimicrobial agents such as essential oils and most antibiotics have hydrophobic character and can easily penetrate Gram-positive bacteria cell walls because this type of bacterium is rich in mucopolysaccharides and protein and low in phospholipids (15,38). The lipopolysaccharides (LPS) present on the surface of the outer membrane of Gram-negative bacteria have a repellent effect on the hydrophobic components of EO, hindering its entry through the cell wall. However, in a study by Dorman and Deans (47), carvacrol and thymol were able to cause disintegration of the outer membrane of Gram-negative bacteria, releasing lipopolysaccharides (LPS) and increasing the permeability of the cytoplasmic membrane to ATP (48).
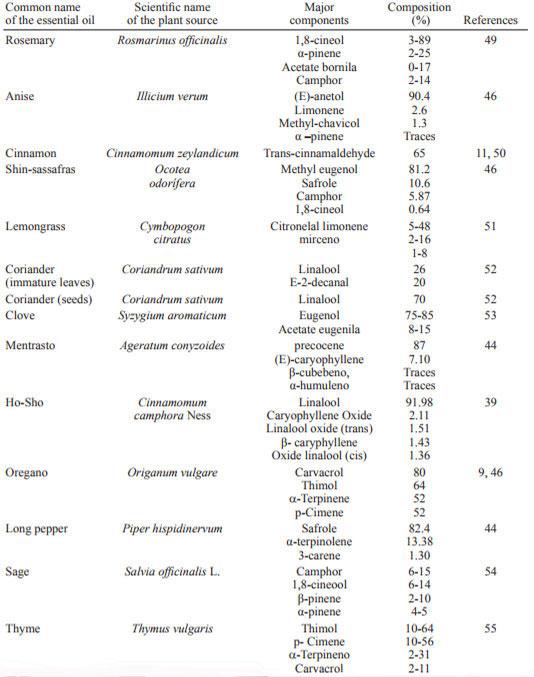
The presence of divalent ions such as Mg2+in LPS will increase the crosslink between molecules, thereby reducing the pore size and further limiting the passage of bioactive compounds (7). Table 4 shows the principal essential oils with microbial activity described in the literature.
Table 4. Main essential oils with antimicrobial activity
The inappropriate use of antimicrobials results in the selection and the consequent spread of resistant strains of various microorganisms (27). The emergence of resistance of microorganisms such as Salmonella to antimicrobial drugs constitutes a double threat to humans and animals, due to the intense use of antimicrobials in human and veterinary medicine, requiring the use of other products that prevent the multiplication of microorganisms. The essential oils from oregano, thyme and cinnamon, among others, have significant antimicrobial potential (11).
In low-acid mayonnaise, the use of essential oils can be regarded as an additional obstacle to the survival and proliferation of Salmonella. However, their use in foods for preservation is limited due to the intense flavor and odor they give when used at effective doses. The addition of oregano essential oils (thymol and isotimol) at a sensorially acceptable concentration of 0.70% is a natural alternative that contributes to the intrinsic safety of mayonnaise, acting synergistically with low pH and low storage temperature (27).
Conclusion and future perspectives
Lipid oxidation in food products, along with the growth of undesirable microorganisms, results in the development of rancidity and spoilage, making the products unacceptable for human consumption. This review analyzed important factors affecting lipid oxidation and microbial growth in mayonnaise. With the monitoring of these parameters, it is possible to slow the lipid oxidation and increase the shelf life of low-acid mayonnaise. Other strategies to prevent oxidation should also be considered, such as reducing the concentration of oxygen in food (vacuum packaging) and decreasing the storage temperature. However, the exclusion of oxygen in a food is difficult, so one of the most effective means of delaying lipid oxidation in mayonnaise is to incorporate antioxidants.
Parallel to this, to prevent microbial contamination of low-acid mayonnaise, effective measures are the use of pasteurized egg yolk, refrigerated storage, modified atmosphere (carbon dioxide), control of acidity in the aqueous phase, proper choice of acid used, control of pH, type and quantity of used, and educational and informational measures directed to the preparation of the product, among others.
Similarly, the sale of mayonnaise at room temperature is an aspect that needs to be reviewed in Brazil by the National Health Surveillance Agency. Refrigeration would minimize the possible occurrence of contamination of egg yolks by Salmonella spp..
The use of essential oils in phytotherapy is related to activities of secondary metabolites, which have antimicrobial, spasmolytic, antiviral and anti-carcinogenic activities, among others. In addition, many essential oils and isolated compounds of them have recently been recognized as powerful natural antioxidants, which could be used as potential substitutes for synthetic antioxidants.
In relation to conventional control methods employing synthetic antimicrobials and antioxidants with broad spectrum of action, the use of natural products such as essential oils stands out by showing good results.
As people become more concerned with their health, there is a global trend of using natural antioxidants and antimicrobials in food products. Several species can be used as sources of natural antioxidants and antimicrobials.
However, more studies need to be performed to evaluate the efficacy and safety of these products. In the particular case of mayonnaise, since contains a variety of different components, there is still a lack of knowledge about the influence of these components on the effectiveness of natural resources with antioxidant and antimicrobial activity. In addition, the elucidation of the mechanism of oxidation in mayonnaise and a better understanding of action and efficacy of such natural products with antioxidant and antimicrobial activity is of great technological importance to the food industry.
Acknowledgements
We thank the Office for the Improvement of Higher Education Personnel (CAPES) of the Ministry of Education, the Rio de Janeiro State Research Foundation (FAPERJ) and Embrapa Food Technology for their financial support.
References
- Gorji SG, Smyth HE, Sharma M, Fitzgerald M. Lipid oxidation in mayonnaise and the role of natural antioxidants: a review. Trends Food Sci Tech, 2016; 56: 88-102.
- Kishk YFM, Elsheshetawy HE. Effect of ginger powder on the mayonnaise oxidative stability, rheological measurements, and sensory characteristics. Ann Agric Sci, 2013; 58 (2): 213-220.
- Mattia C, Balestra F, Sachetti G, Neri L, Mastrocola D, Pittia P. Physical and structural properties of extra-virgin olive oil based mayonnaise. Food Sci Technol-LEB, 2015; 62 (1): 764-770.
- Elias SO, Tomasco PV, Alvarenga VO, Sant’ana AS, Tondo EC. Contributor factors for the occurrence of salmonellosis during preparation, storage and consumption of homemade mayonnaise salad. Food Res Int, 2015; 78: 266-273.
- Malheiros OS, De Paula CMD, Tondo EC. Cinética de crescimento de Salmonella Enteritidis envolvida em surtos alimentares no RS: uma comparação com linhagens de outros sorovares. Ciênc e Tecnol de Aliment, 2007; 27 (4):751-755.
- Masuchi MH, Celeghini RMS, Gonçalves LAG, Grimaldi R. Quantificação de TBHQ (Terc Butil Hidroquinona) e avaliação da estabilidade oxidativa em óleos de girassol comerciais. Quím Nova, 2008; 31 (5): 1053-1057.
- Danneberg GS, Funck GD, Mattei FJ, Silva WP, Fiorentini, AM. Antimicrobial and antioxidant activity of essential oil from pink pepper tree (Schinus terebinthifolius Raddi) in vitro and in cheese experimentally contaminated with Listeria monocytogenes. Innov Food Sci and Emerg Tech, 2016; 36: 120-127.
- Luca ANB, Koerich GMD. Perfil Epidemiológico dos Surtos de DTA Causados por Salmonella sp. em Santa Catarina, Brasil, Notificados no SINAN NET de 2006 A 2008. 2009. 20 f. Monografia (Especialização) - Curso de Especialização em Microbiologia, Departamento de Microbiologia, Pontifícia Universidade Católica do Paraná, Curitiba, 2009.
- Elias SO. Modelagem dos Parâmetros Cinéticos de Multiplicação de Salmonella Enteritidis SE 86 em Maionese Caseira e Práticas de Preparo, Estocagem e Consumo desse Alimento no Rio Grande do Sul. 2014. 104 f. Dissertação (Mestrado) - Programa de Pós Graduação em Microbiologia Agrícola e do Ambiente, Universidade Federal do Rio Grande do Sul, Porto Alegre, 2014.
- Nakatani N. Phenolic antioxidants from herbs and spices. Biofactors, 2000; 13: 141-146.
- Santurio JM, Santurio DF, Pozzatti P, Moraes C, Franchini PR, Alves SH. Atividade antimicrobiana dos óleos essenciais de orégano, tomilho e canela frente à sorovares de Salmonella enterica de origem avícola, Ciênc Rural, 2007; 37 (3): 803-808.
- Bakkali F, Averbeck S, Averbeck D. Biological effects of essential oils – A review. Food Chem Toxicol, 2008; 46 (2): 446-475.
- Amorati R, Foti MC, Valgimigli L. Antioxidant activity of essential oils. J Agric Food Chem, 2013; 61: 10835-10847.
- Lahlou M. Methods to study the phytochemistry and bioactivity of essential oils. Phytother Res, 2004; 18: 435-448.
- Bouchekrit M, Laouer H, Hajji M, Nasri M, Haroutounian SA, Akkal S. Essential oils from Elaeoselinum asclepium: Chemical composition, antimicrobial and antioxidant properties. Asian Pac J Trop Biomed, 2016; 6 (10): 851-857.
- Bajpai VK, Baek KH, Kang SC. Control of Salmonella in foods by using essential oils: A review. Food Res Int, 2012; 45: 722-734.
- Sánchez-Maldonado AF, Schieber A, Ganzle MG. Structure-function relationships of the antibacterial activity of phenolic acids and their metabolism by lactic acid bacteria, J Appl Microbiol, 2011; 111: 1176-1184
- Ramalho VC, Jorge N. Antioxidantes utilizados em óleos, gorduras e alimentos gordurosos. Quím Nova, 2006: 29 (40): 755- 760.
- Boroski M, Giroux H, Sabik H, Petit HV, Visentainer JV, Pintro PTM, Britten NM. Use of oregano extract and oregano essential oil as antioxidants in functional dairy beverage formulations. Food Sci Technol-LEB, 2012; 47: 167-174.
- Robles-Ramírez MC, Monterrubio-López R, Mora-Escobedo R, Beltrán-Orozco MC. Evaluation of extracts from potato and tomato wastes as natural antioxidant additives. Arch Latinoam Nutr, 2016; 66 (1): 66-73.
- Jaeger J. Produção de Maionese. 2012. 160 f. Trabalho de Conclusão de Curso (Engenharia Química) - Curso de Ciências Tecnológicas, Universidade Regional de Blumenau, Blumenau, 2012.
- Amin MHH, Elbeltagy AE, Mustafa M, Khalil AH. Development of low fat mayonnaise containing different types and levels of hydrocolloid gum. J Agroaliment Proc Technol, 2014; 20 (1): 54-63.
- Brasil. MINISTÉRIO DA SAÚDE. AGÊNCIA NACIONAL DE VIGILÂNCIA SANITÁRIA. Resolução nº 276, de 22 de setembro de 2005. Aprova o Regulamento Técnico para Especiarias, Temperos e Molhos. Diário Oficial [da República Federativa do Brasil], Brasília, DF, 22 de setembro de 2005.
- Food and Drug Administration. CFR - Code of Federal Regulations Title 21, PART 169 -FOOD DRESSINGS AND FLAVORING. Subpart B-Requirements for Specific Standardized Food Dressings and Flavorings. Sec. 169.140 Mayonnaise, 1993.
- Silva JPL, Souza EF, Modesta RCD, Gomes IA, Silva OF, Franco BDGM. Antibacterial activity of nisin, oregano essential oil, EDTA, and their combination against Salmonella Enteritidis for application in mayonnaise. Vigil Sanit Debate, 2016; 4 (1): 83-91.
- Rodrigues ML. Azeite de pequi: efeito do aquecimento em temperatura de fritura e utilização como ingrediente na formulação de maionese. 2011. 94 p. Dissertação (Mestrado) - Escola de Agronomia e Engenharia de Alimentos da Universidade Federal de Goiás, Goiás, 2011.
- Téo CRPA, Oliveira TCRM. Salmonella spp.: O ovo como veículo de transmissão e as implicações da resistência antimicrobiana para a saúde pública. Cienc Agrar, 2005; 26 (2): 195-210.
- Gole VC, Roberts JR, Sexton M, May D, Kiermeier A, Chousalkar KK. Effect of egg washing and correlation between cuticle and egg penetration by various Salmonella strains. Int J Food Microbiol, 2014; 18: 182–183.
- Jay JM. Modern Food Microbiology, 2000. 6 ed. Local de publicação: Aspen Publishers, 625 p.
- Araújo JMA. Química de Alimentos: Teoria e Prática, 1995. Viçosa-MG: Imprensa Universitária, 355p.
- Martin G. Comportamento de Salmonella em ovo em pó em função da Atividade de água (Aa) e do binômio Tempo x Temperatura de armazenamento. 2005. 81 p. Dissertação (Mestrado). Departamento de Alimentos e Nutrição Experimental, Faculdade de Ciências Farmacêuticas da Universidade de São Paulo, São Paulo, 2005.
- Mattos DA, Araújo ES, Aragão SF, Fook SML, Vieira KVM, Meira CMBS, Santiago AM. Qualidade dos grãos de milho utilizados em uma indústria alimentícia de Campina Grande - PB, no período de 2004-2005. Rev Bras Toxicol, 2009; 22 (1-2): 34-41.
- Ditchfield C. Estudo dos métodos para medida da atividade de água. 2000. 195 p. Dissertação (Mestrado). Escola Politécnica, Universidade de São Paulo, São Paulo, 2000.
- Depree JA, Savage GP. Physical and flavour stability of mayonnaise. Trends Food Sci Tech, 2001; 12 (5): 157-163
- acobsen C, Timm M, Meyer AS. Oxidation in fish oil enriched mayonnaise: Ascorbic acid and low pH increase oxidative deterioration. J Agric Food Chem, 2001; 49 (8): 3947-3956.
- Embuscado ME. Spices and herbs: Natural sources of antioxidants – a mini review. J Funct Foods, 2005; 18: 1-10.
- Kulisic T, Radonic A, Milos M. Inhibition of lard oxidation by fractions of different essential oils. Grasas Aceites, 2005; 56: 284-291.
- Cansian RL, Mossi AJ, Oliveira D, Toniazzo G, Treichel H, Paroul N, Astolf V, Serafini LA. Atividade antimicrobiana e antioxidante do óleo essencial de ho-sho (Cinnamomum camphora Ness e Eberm Var. Linaloolifera fujita). Rev Ciênc Tecnol, 2010; 30 (2): 378-384.
- Lagouri V, Boskou D. Nutrient antioxidants in oregano. Int J Food Sci Nutr, 1996; 47: 493-497.
- Roesler R, Malta LG, Carrasco LC, Holanda RH, Sousa CAS, Pastore GM. Atividade antioxidante de frutas do cerrado. Ciênc Tecnol Aliment, 2007; 27 (1): 53-60.
- Mimica-Dukic N, Bozin B, Sokovic M.; Simin M. Antimicrobial and antioxidant of Melissa officinalis L. (Laminaceae) essential oil. J Agric Food Chem, 2004; 52 (9): 2485-2489.
- Vicentino ARR, Menezes FS. Atividade antioxidante de tinturas vegetais, vendidas em farmácias com manipulação e indicadas para diversos tipos de doenças pela metodologia de DPPH. Rev Bras Farmacogn, 2007; 17 (3): 384-387.
- Bozin B, Mimica-Dukic N, Simin N, Anackov G. Characterization of the volatile composition of essential oils of some Lamiaceae spices and the animicrobial and antioxidant activities of the entire oils. J Agric Food Chem, 2006; 54 (5): 1822-1828.
- Calo JR, Crandall PG, O’Bryan CA, Ricke SC. Essential oils as antimicrobials in food systems – A review. Food Control, 2015; 54: 111-119.
- Hajji M, Masmoudi O, Souissi N, Triki Y, Kammoun S, Nasri M. Chemical composition, angiotensin I-converting enzyme (ACE) inhibitory, antioxidant and antimicrobial activities of the essential oil from Periploca laevigata root barks. Food Chem, 2010; 121 (3): 724-731.
- Lima RK, Cardoso MG, Moraes JC, Carvalho SM, Melo BA, Vieira SS. Composição química e toxicidade de óleos essenciais para o pulgãoverde Schizaphis graminum (Rondani, 1852). Arq Inst Biol, 2014; 81 (1): 22-29.
- Dorman HJD, Deans SG. Antimicrobial agents from plants, antibacterial activity of plant volatile oils. J Appl Microbiol, 2000; 88: 308-316.
- Burt S. Essential oils: their antibacterial properties and potential applications in foods-a review. Int J Food Microbiol, 2004; 94: 223– 253.
- Daferera DJ, Ziogas BN, Polissiou MG. The effectiveness of plant essential oils in the growth of Botrytis cinerea, Fusarium sp. and Clavibacter michiganensis subsp. michiganensis. Crop Prot, 2003; 22 (1): 39-44.
- Lens-Lisbonne C, Cremieux A, Maillard C, Balansard G. Methodes d’evaluation de l’activite´ antibacterienne des huiles essentielles: application aux essences de thym et de cannelle. J Pharm Belg, 1987; 42 (5): 297-302.
- Hammer KA, Carson CF, Riley TV. Antimicrobial activity of essential oils and other plant extracts. J Appl Microbiol, 1999; 86: 985-990.
- Delaquis PJ, Stanich K, Girad B, Mazza G. Antimicrobial activity of individual and mixed fractions of dill, cilantro, coriander and eucalyptus essential oils. Int J Food Microbiol, 2002; 74: 101-109.
- Bauer K, Garbe D, Surburg H. Common Fragrance and Flavor Materials: Preparation, Properties and Uses. Wiley-VCH, 923, 2001.
- Marino M, Bersani C, Comi G. Impedance measurements to study the antimicrobial activity of essential oils from Lamiacea and Compositae. Int J Food Microbiol, 2001; 67: 187-195.
- Juliano C, Mattana A, Usai M. Composition and in vitro antimicrobial activity of the essential oil of Thymus herba-barona Loisel growing wild in Sardinia. J Essent Oil Res, 2000; 12: 516–522.