Introduction
The functional food market is expanding due to rising demand for digestive and Immune health products, particularly prebiotics, probiotics, and postbiotics (PPPs) (1).
The prebiotic health market (USD 2.99B in 2024) is driven by inulin use in dairy and infant formula, with a 5.15% Compound Annual Growth Rate (CAGR) (2024–2029) (2). Probiotic (USD 113.43B by 2029, CAGR: 7.53%) dominate Asia-Pacific, offering gut and immune benefits (3, 4) and is the fastest-growing region, with probiotic products available in 196 countries (2, 5). Postbiotics (e.g., microbial metabolites) gain traction for stability and clinical applications (6). This study analyzes regulatory gaps in LAC’s PPP market using Asymmetric Information Theory (7,8,9), highlighting: misleading claims from producer-consumer information gaps, lagged standards vs. global frameworks (WHO, EFSA) and policy needs: transparent labeling, verified health claims, and safety protocols. Methods include a literature review (2010–2025, Google Scholar/Scielo) and internet market analysis of LAC distribution channels. In this analysis, information on prebiotics, probiotics, and postbiotics is analyzed for each continent and country only in the market where information on them was found.
Theoretical and Legal foundations review in the commercialization of the PPPs products in international markets PPPs regulation faces a “market for lemons” risk where low-quality products may substitute high-quality ones due to information asymmetry. Manufacturers exploit regulatory gaps by lowering standards post-market, while consumers struggle to verify label claims about strains and concentrations (10).
The functional food market addresses this through certifications, scientific studies and traceability technologies like blockchain. The Pan American Health Organization defines functional foods as containing bioactive components that offer health benefit beyond nutrition (10), supporting the shift from “adequate” to “optimal” nutrition (11). These foods must demonstrate safety and efficacy at normal consumption levels, though regulatory rigor varies globally.
Prebiotics (inulin, Fructooligosaccharides (FOS), Galactooligosaccharides (GOS) selectively nourish beneficial gut bacteria (12-13). While WHO/FAO (14) and EFSA provide guidelines, regulatory harmonization remains challenging.
Marketed in infant foods (improving stool quality and gut flora) (15), they now include innovative formats like pectin-derived oligosaccharides (POS) (16). However, high R&D costs and strict labeling requirements in developed countries hinder market growth.
Comparative international regulation PPPs food and distribution channels in the world Prebiotic food in the world and market and regulatory authority Prebiotic production has expanded significantly due to gut health awareness (17). Inulin, FOS, and GOS are widely used in functional foods, supported by evidence of microbiotabenefits (18-19). Market share leaders: Asia-Pacific (44%), North America (28%), Europe (19%) (20). EFSA/FDA approvals drive innovation (21), but cost and education challenges persist (22).
Asia Prebiotic Market
Asia Pacific’s feed prebiotics market grows due to antibiotic restrictions, with India’s FSSAI implementing new veterinary drug rules (2025) (23). Indonesia’s National Agency of Food Control BPOM approved “prebiotic” for chicory-root inulin/oligofructose (≥4.5g/L, 30:70 ratio) in milk powder for ages 3+ (24), reflecting demand for digestive health solutions.
North America Prebiotic Market
United States: FDA regulates PPPs as food additives (21 CFR 172), GRAS substances (25), and dietary supplements (DSHEA) (26), requiring clinical studies for health claims (27). Canada classifies prebiotics as functional foods/NHPs under Food and Drug Regulations (28).
Europe Prebiotic Market- European Commission (EC)– Directorate General for Health and Consumers. Directive on Food Supplements (Directive 2002/46/ EC)
The EU regulates prebiotics under multiple frameworks: food supplements must meet Safety standards (29), Regulation (EU) No. 432/2012 authorizes EFSA-backed health claims (31-32), and novel foods require safety assessments (34). Key points: “prebiotic” claims are unauthorized (30,35); approved ingredients like GOS and 3-Fucosyllactose (3-FL) undergo strict evaluations (33-34). Regulation (EC) No 1924/2006 governs claims (33). Market leaders include Tereos Group and Royal Cosun (2). EFSA approves specific claims (e.g., gut health) but requires strong evidence (32,35). Recent approvals expand GOS uses (33-34).
Probiotic Food in the World and Market
Probiotics, live microorganisms providing health benefits when consumed adequately,
Have evolved from niche supplements to mainstream products. Growing awareness of the Gut microbiome’s health role has driven scientific and commercial interest. Research Supports their benefits for gut health, immunity, and mental well-being (35,36).
FAO/WHO established probiotic evaluation guidelines in 2002 (37). Market leaders: Asia Pacific (36%), North America (25%), Europe (20%), LAC (15%), and MEA (5%). This growth reflects strong scientific validation and consumer demand for microbiome-supporting products.
North America Probiotic Market.
USA – FDA and Canada- HC. Food Directorate
It is possible to consider “functional foods” in any of the aforementioned food and supplement categories, based on their natural origin. From a legislative perspective, foods with probiotics could fit into several of the four food categories described by the FDA; however, in the United States, the health benefits of dairy products with added probiotics, prebiotics, or cultures are not explicitly recognized. The main probiotic strains in USA are: L. acidophilus. Source: Rhodia, Inc. (Madison, WI. USA.); L. acidophilus DDS-1. Source: Nebraska Cultures, Inc. (Lincoln, NE. USA); L. crispatus CTV05. Source: Gynelogix, (Boulder, CO, USA) and S. boulardii. Source: Biocodex Inc. (Seattle, WA. USA).
In Canada are: L. fermentum RC-14. Source: Urex Biotech Inc. (London, Ontario, Canada); L. acidophilus R0011. Source: Institut Rosell (Montreal, Canada) (38).
European Union Probiotic Market
Global definitions of functional foods vary: Europe/U.S. classify them as GRAS, while LAC countries regulate them as supplements. Prebiotic/probiotic claims differ; postbiotics
Show promise for gut health (39). Key strains include L. acidophilus LA-1 (Chr. Hansen), L. rhamnosus GG (Valio Dairy), and others (38). Regulatory gaps persist, urging Standardized frameworks. Postbiotics gain traction for stability and clinical benefits.
Asia and Australian Prebiotic /Probiotic Market
China (MOH): Prebiotics/probiotics fall under multiple categories (40). GOS/FOS are fortifiers; inulin—novel food; lactulose—additive. “Prebiotic” claims restricted in infant foods. Approval: 1–2 years. Japan (MHLW/ CAA): Regulates via FOSHU (41,42) & FFC (43). FFC updates mandate GMP by 2026 (43). Key strain: L. casei Shirota (Yakult) (38). Australian continent (FSANZ/TGA): Guides probiotics/ postbiotics in medicines (44). Market strain: B. lactis HN019 (38).
Latin America and the Caribbean - (LAC) Prebiotic/ Probiotic Market
Latin America regulates prebiotics under varied frameworks, often overlapping with probiotics. Argentina (ANMAT): Functional ingredients/additives under Food Code Articles1383 (approved prebiotics) & 1385 (claims) (45), plus Disposition 4980/2005 for Functional foods (46). Brazil (ANVISA): Classified as functional ingredients/additives under RDC16/1999 (48), RDC 18/2008 (scientific proof required) (49), and IN 60/2016 (Approved claims) (50). Chile: Prebiotics are additives (RSA Art. 120) (51,52) or supplements (Decree 309/2015) (52) but lack legal definition (53). Claims require evidence (54), facing classification challenges (55). Market includes products like NUP! Pylori OFF (56). Colombia (INVIMA): Functional ingredients under Res. 333/2011 (health benefits) (58) And 810/2021(labeling) (59). Mexico (COFEPRIS): Regulated as Functional ingredients/ additives under NOM-086 (functional foods) (60) and NOM-218 (supplements) (61), requiring premarket approval (62). Peru (DIGESA): Governed by Supreme Decree 007-98-SA (safety) (63) and Resolution 546-2013 (claims) (64), mandating scientific evidence. Regional challenges include inconsistent definitions despite growing demand.
Postbiotic Food in the World and Market
Postbiotics, derived from probiotic activity, enhance gut health (65) and immune function.
The global market is growing at 12-15% CAGR (2023-2030) due to gut health awareness and postbiotic stability (66)(67). North America (15-20% share): Largest market (U.S./Canada), led by ADM(USA)and Lallemand (Canada (2, 68). Europe (25-30% share): Strongin Germany/ France, with Nestlé (Switzerland) and Danone (France) leading (67) (69). Asia-Pacific (40-45% share): Fastest-growing (Japan/China/ India), driven by Morinaga Milk (Japan) (70). LAC (5%) & MEA (2%): Emerging markets (71). Applications: Supplements (40%), functional foods (30%), infant nutrition (15%), animal feed (10%) (72). Advantages: Shelf stability, proven efficacy (73, 74). Regulatory approvals (FDA/ EFSA/FSSAI) boost adoption (75).
Methodology in the normative economic analysis of food regulations
This study employs a sequential mixed-methods design, utilizing DCA to examine food regulatory frameworks (75,76) as the primary methodology and as a complementary analytical technique, the SLR was conducted following PRISMA guidelines, with a protocol registered in PROSPERO that defined PICOS/S inclusion/exclusion criteria. The earch across key databases utilized controlled vocabulary and AI tools to prioritize relevant records and control for selection bias. The selection process was documented in a PRISMA flow diagram. Evidence quality was assessed using an adapted GRADE approach, integrating Asymmetric Information Theory with economic/ social science concepts. The DCA analyzed the SLR corpus, employing a deductive-inductive coding matrix. Deductive categories integrated Asymmetric Information Theory to identify power structures and the FAIR (Findability, Accessibility, Interoperability, Reusability) and DEIA (Diversity, Equity, Inclusion, and Accessibility) principles to examine discursive treatment. Inductive categories captured emergent themes. The resulting narrative synthesis intertwines the thematic findings from the SLR with the discursive analysis from the DCA. The FAIR/DEIA principles transversally guided methodological ethics, ensuring equity through the inclusion of grey literature and bias mitigation. Thus, this framework critically articulates “what is known” (SLR) with “how it is stated and what relations are configured” (DCA). The approach involves: (1) reviewing prior studies; (2) systematically analyzing regulatory documents; and (3) organizing data into three themes: food safety policies, product/market inspections, and manufacturing/labeling rules. Data sources include Google Scholar/Scielo (2010–2025), comparing Latin American (LAC) and international standards. DCA follows four phases: document selection, logical analysis, scientific abstraction, and multi-source synthesis (bibliographic/digital/institutional). Systematic Literature Review (SLR) principles enhance rigor (77), with an 8-step qualitative design. Step 5 includes: academic searches, snowballing, and gray literature collection. The protocol begins with: Stage 1 PICOS-guided research questions: “What regulatory advances exist in PPPs commercialization in LAC (2010–2025) vs. North America/Asia/Europe?”. Implementation: Stage 2 applied the SLR protocol (78), analyzing LAC regulations via Scielo/Google Scholar and GEMINI AI (English/ Spanish/Portuguese). Keywords: “pre/pro/postbiotics,” “regulations,” “LAC” (Table 1). Cultural contexts were addressed, focusing on three regulatory dimensions with narrative synthesis. Data Retrieval: Stage 3 extracted data from Google Scholar (global) and Scielo (LAC-specific), using: (1) database searches, (2) snowballing, and (3) gray literature. Screening followed FAIR/DEIA principles: abstract review → full- text analysis. No yield limits were set; gray literature supplemented peer-reviewed sources.
Table 1. Search equations for PPP food in Human Health

Source: Own elaboration, 2025
Test evaluation and analysis
Research was evaluated using: publication metrics (year/ location); GRADE system (social science adaptation); 9-point relevance scale. Narrative synthesis compared evidence quality/quantity, employing three legislative-focused qualitative codes to streamline analysis. Methodological choices balanced rigor with resource optimization in regulatory literature examination.
Results
The analysis of PPPs literature reveals a predominant focus on nutritional, medical, and biotechnological aspects rather than legal/regulatory frameworks. Despite using Scielo and Google Scholar - comprehensive databases for LAC publications in Spanish, English, and Portuguese - minimal documentation exists on PPPs regulations in the region. The study examined basic PPPs commercialization rules, tracking regulatory specifications and their evolution across countries. Methodological consistency was maintained through systematic citation management and comprehensive content analysis (beyond abstracts). Findings indicate ongoing global disagreement about defining functional foods/PPPs, with Economic organizations adopting varied conceptualizations.
While some nations update
Food regulations with technological advances, others resist adoption of PPPs, resulting in Scarce publications about regional regulatory progress.
LAC literature primarily addresses PPPs conceptualization, innovation, and applications rather than legislative aspects. Both databases show limitations: articles typically discuss general food characteristics or health/nutrition aspects without consistent legislative or public policy approaches. Google Scholar provided information across all three established search dimensions (food safety policies, product/market inspections, manufacturing/labeling), while Scielo mainly covered dimension 1 (food safety policies). This regulatory gap persists despite PPPs’ growing commercial importance, highlighting the need for standardized definitions and region-specific legal frameworks that address both technological advancements and public health considerations. In both databases, bibliographic reviews reveal a paucity of peer-reviewed articles on the topic, as well as comparative studies between similar products in various Latin American topic, as well as comparative studies between similar products in various LAC countries. The results indicate a lack of information on the legislative and marketing management of PPP food products. What they do share individually as a country and collectively is the use of the Codex Alimentarius. The initial results of the analysis in the Scielo database for the areas of social sciences, health, multidisciplinary and law, are shown in Table 2. Regulations on the types of foods addressed are more related to product quality controls and their production certifications for marketing—such as ISO 9000 or HACCP— than to the identification of their content, nutrients, or special or differentiating characteristics. The Scielo database has indexed peer review; however, the Google Scholar database, although it finds a greater number of articles published in the three dimensions of the study indicated, the types of arbitration for their publication are different. Analysis reveals international PPPs regulations focus on certification, while LAC lacks value-based standards. Most cover only general food safety rules. Scielo yielded just one article (Dimension 1), versus Google Scholar’s 14 (6-D1, 3-D2, 5-D3). See Table 2. Developed Nations (Japan/US/EU) have advanced PPPs frameworks, while LAC relies on Codex Alimentarius (WHO/FAO/WTO). Argentina, Brazil, Mexico, Chile, and Colombia dominate regional PPPs literature, but lack legislative proposals. Unlike US/Asia/Europe, no regulatory progress exists in LAC, with minimal research on policy changes for PPPs.
Table 2. Scielo and Google Scholar Database Search of PPPs - LAC 2010-2025
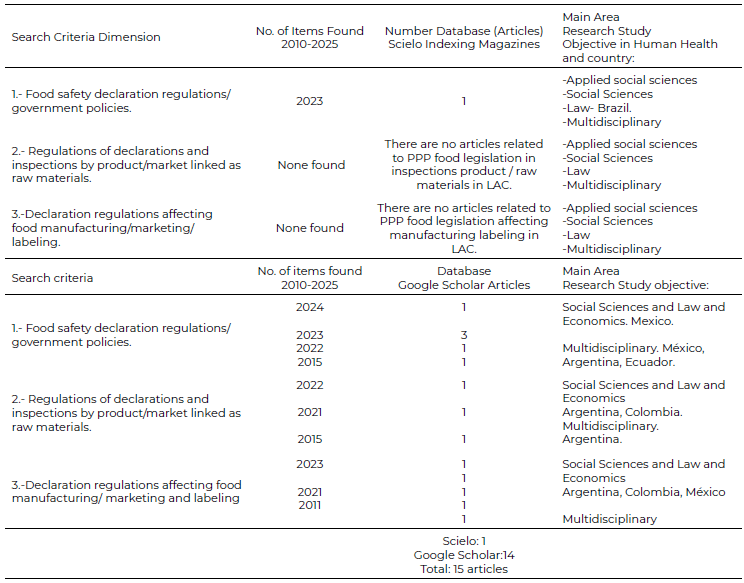
Source: Own elaboration, 2025
The study identified a small number of articles on regulatory aspects, partly due to the nature of the platforms consulted. Google Scholar and Scielo have a broad and universal scope, with a strong emphasis on scientific literature produced mainly in Latin America and the Caribbean, Spain, and Portugal, where regulatory debate is limited, and it was precisely for this reason that they were selected. The results indicate that there is global disagreement regarding the definition of functional foods/PPP, and that economic organizations adopt diverse conceptualizations.
Methodological consistency was maintained through systematic citation management and in-depth content analysis. This shows that developed countries update food regulations in line with technological advances, while developing countries only adopt PPPs, resulting in few publications on the evolution of regional regulations. The literature from Latin America and the Caribbean mainly addresses the conceptualization, innovation, and applications of PPPs, but not the legislative aspects of public policies.
Policy implications LAC and global markets show a legislative gap for PPPs regulation, with more research in nutrition/biotechnology than policy. Analysis focused on: (1) food safety policies, (2) product/ market inspections, and (3) labeling rules. PPPs lack a universal definition but include health-enhancing foods. As a growing market segment, they require specific regulations and public health integration.
This study implemented a sequential mixed-methods framework to analyze regulatory frameworks for PPP products in LAC. The systematic review, conducted following PRISMA guidelines with a protocol registered in PROSPERO, identified and evaluated evidence on regulatory developments (2010-2025). The use of controlled vocabulary, AI tools (GEMINI AI), and an adapted GRADE approach integrating asymmetric information theory minimized selection biases. The inclusion of gray literature following FAIR/ DEIA principles incorporated underrepresented perspectives. The DCA employed a deductive-inductive coding matrix. Deductive categories, based on asymmetric information theory and FAIR/DEIA principles, deconstructed regulatory discourse to identify related structures. The inductive categories captured emerging themes. The narrative synthesis integrated the thematic findings of the SLR (food safety policies, inspections, and labeling regulations) with the discursive analysis of the DCA. This approach confirmed a critical regulatory gap in Latin America and the Caribbean, characterized by dependence on the Codex Alimentarius and the predominance of technoscientific approaches over normative ones. The study demonstrates that addressing this gap requires not only legislative proposals but also a discursive shift that prioritizes regulatory sovereignty, equity, and transparency.
Discussion
The obtained results confirm the existence of a critical gap in the regional scientific Literature concerning the regulatory framework for PPPs. This discussion is structured around three main axes derived from the findings: First, the dominance of techno-scientific approaches over regulatory ones. The overwhelming preference for research on nutritional, medical, and biotechnological aspects reflects a global trend in PPP literature.
However, within the LAC context, this disparity is more acute, marginalizing legal and public policy research. This suggests that technological innovation and scientific research are advancing at a much faster pace than the regulatory capacity of countries in the region. The direct consequence is a legal vacuum that can hinder intra-regional commercialization, fail to protect consumers, and stifle R&D investment by creating an environment of legal uncertainty. Second, the dependence on the Codex Alimentarius and the lack of regulatory sovereignty. The finding that LAC countries share an almost exclusive use of the Codex Alimentarius as a normative reference is a double-edged sword. While it provides a harmonized and internationally recognized foundation, it also highlights a lack of endogenous regulatory development tailored to the region’s specific realities. The frameworks of Japan, the U.S., and the EU are proactive and integrated with their innovation and public health policies. In contrast, Latin America’s reliance on general Codex standards, often focused on safety and quality certifications (ISO, HACCP), leaves crucial aspects of PPPs unaddressed, such as health claims, labeling veracity, and definition standardization. This lack of “regulatory sovereignty” places the region in a reactive position and makes it vulnerable to importing foreign regulations that may not be suitable. Third, the limitations of databases and the publication ecosystem. The methodological choice to use Scielo and Google Scholar was intentional to capture the intellectual production of LAC, but the results also expose the inherent limitations of this publication ecosystem. The scarcity of peer-reviewed articles on legislative aspects is not merely a search problem but a symptom of a larger research deficit. The predominance of grey literature and the lack of in-depth comparative studies indicate that regulatory knowledge may be dispersed in government reports, theses, or unindexed technical documents, making access and systematization difficult.
Furthermore, Google Scholar’s superiority in retrieving information across all dimensions underscores its utility for comprehensive searches on emerging topics where formal literature is scarce, although it requires more rigorous screening. This translation maintains a formal American English academic tone, using precise terminology and complex sentence structures appropriate for scholarly discourse. It faithfully conveys the original Spanish meaning while adhering to conventions of English academic writing.
Acknowledgments
We appreciate the financial support provided by the Catholic University of Temuco for the publication of this article. We also thank the experts who validated the relevance and qualitative content of this material.
Conflict of Interest
The authors declare they have no financial interests or personal relationships that could have influenced the work presented in this article. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- Mordor Intelligence. Tamaño de mercado de ingredientes prebióticos y análisis de acciones.2025. https://www.mordorintelligence.com/es/industry-reports/prebiotic-ingredient-market
- Mordor Intelligence. Tamaño de mercado de prebióticos para piensos. Análisis de tamaño y participación. Tendencias de crecimiento y pronósticos 2029. 2025. https://www.mordorintelligence.com/es/industry-reports/global-prebiotics-market-industry.
- Gul S, Durante-Mangoni E. Unraveling the Puzzle: Health Benefits of Probiotics-A Comprehensive Review. J Clin Med 2024;13(5):1436. https://doi.org/10.3390/jcm13051436
- Mordor Intelligence. Mercado de probióticos - Tamaño, análisis, crecimiento de la industria y tendencias. 2025. https://www.mordorintelligence.com/es/industry-reports/probiotics-market
- World Gastroenterology. Probióticos y prebióticos. 2023. https://www.worldgastroenterology.org/guidelines/probiotics-and-prebiotics/probiotics-and-prebiotics-spanish
- Salminen S, Collado MC, Endo A, Hill C, Lebeer S, Quigley EM, et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat Rev Gastroenterol Hepatol. 2021;18(9):649–67. https://doi.org/10.1038/s41575-021-00440-6
- Akerlof, G. A. The market for “Lemons”: Qualitative uncertainty and the market mechanism, 1970; 86 (3): 488-500. https://doi.org/10.2307/1879431
- Spence, M. Product Selection, Fixed Costs, and Monopolistic Competition, The Review of Economic Studies, 1976; 43(2): 217–235. https://doi.org/10.2307/2297319
- Stiglitz, J. Incentives and Risk Sharing in Sharecropping. Rev. Econ. Stud.1974; 41 (2): 219-255. https://doi.org/10.2307/2296714
- Organización Panamericana de la Salud (OPS). Reducción de la sal. 2025. https://www.paho.org/es/temas/reduccion-sal.
- Ponomarev S, Mack I, Chouker A. Editorial: Microbiota, nutrition, and stress: Modulators of immunity. Front. Nutr. 2023; 10: 1328346. https://doi.org/10.3389/fnut.2023.1328346
- Gibson GR, Roberfroid MB. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J Nutr 1995; 125(6): 1401-1412. https://doi.org/10.1093/jn/125.6.1401
- Slavin J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients, 2013; 5(4): 1417-1435. https://doi.org/10.3390/nu5041417
- World Health Organization & Food and Agriculture Organization. Guidelines for the evaluation of probiotics in food.2007. https://isappscience.org › probiotic guidelines
- Yoo S, Jung SC, Kwak K, Kim JS. The Role of Prebiotics in Modulating Gut Microbiota: Implications for Human Health. Int J Mol Sci 2024;25(9):4834. https://doi:10.3390/ijms25094834.
- Gullón B. Gómez M. Martínez-Sabajanes, R. Yáñez, J.C. Parajó, J.L. Alonso. Pectic oligosaccharides: Manufacture and functional properties. Trends Food Sci Technol 2013; 30 (2):153-161. https://doi.org/10.1016/j.tifs.2013.01.006.
- Holscher HD. Dietary fiber and prebiotics and the gastrointestinalmicrobiota. Gut Microbes. 2017;8(2):172-184. https://doi.org/10.1080/19490976.2017.1290756
- Jubair HR, Abbas FK, Ali BH, Akool HM, Dhahir TM. Nutritional effects of polyphenols, dietary fiber, prebiotic effect, compounds associated with dietary fiber and impact of polyphenols on human intestinal microbiota and their health benefits. Current Clinical Med Edu 2025; 3(3): 24-38. https://www.visionpublisher.info/index.php/ccme/article/view/217
- Swanson KS, Gibson GR, Hutkins R. et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. 2020. https://doi.org/10.1038/s41575-020-0344-2
- GlobeNewswire. Research and Markets. Global Prebiotics (FOS, Inulin, GOS, MOS) Market Report 2022-2030: Rising Demandin Dietary Supplements in Infant Foods is Expectedto Propel Market Growth.19, 2022. https://www.globenewswire.com/news-release/2022/08/19/2501577/28124/en/Global-Prebiotics-FOS-Inulin-GOS-MOS-Market-Report-2022-2030-Rising-Demand-in-Dietary-Supplements-in-Infant-Foods-is-Expected-to-Propel-Market-Growth.html
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific and technical guidance for the preparation and presentation of an application for authorisation of a health claim (revision 1). EFSA J 2011; 9(5):2170. https://doi.org/10.2903/j.efsa.2011.2170.
- Pandey KR, Naik SR, Vakil BV. Probiotics, prebiotics and synbiotics- a review. J Food Sci Technol. 2015;52(12):7577-7587. https://doi.org/10.1007/s13197-015-1921-1
- Mordor Intelligence. Asia-Pacific Feed Prebiotics Market Size & Share Analysis - Growth Trends & Forecasts (2025-2030). 2025. https://www.mordorintelligence.com/industry-reports/asia-pacific-feed-prebiotics-market-industry.
- Nutraceutical Business Review. Indonesia approves prebiotic claim for inulin and oligofructose. 2025. https://nutraceuticalbusinessreview.com/indonesia-approves-prebiotic-claim-for-inulin-and-oligofructose-176744.
- U.S. Food and Drug Administration. GRAS notices for prebiotic substances. 2018. https://www.fda.gov/food/generally-recognized-safe-gras/gras-notice-inventory.
- National Institutes of Health. Dietary Supplement Health and Education Act of 1994. PublicLaw.103417103rd.Congress. https://ods.od.nih.gov/About/DSHEA_Wording.aspx.
- U.S. Food and Drug Administration. Labeling & nutrition guidance documents ®ulatory information.2019. https://www.fda.gov/food/guidance-documents-regulatory-information-topic-food-and-dietary-supplements/labeling-nutrition-guidance-documents-regulatory-information
- Health Canada. Regulations for natural health products. 2020. https://www.canada.ca/en/health-canada/services/drugs-health-products/natural-non-prescription/regulation.html.
- European Food Safety Authority (EFSA). Safety of extension of use of galacto oligosaccharides (GOS) as a novel food. EFSA J 2021;19(6):6844. https://doi.org/10.2903/j.efsa.2021.6844
- European Commission. Regulation (EU) 2015/2283 on novel foods. OJEU. 2015. https://eur-lex.europa.eu/eli/reg/2015/2283/oj/eng
- European Food Safety Authority. Scientific opinion on the substantiation of health Claims related to prebiotics. 2011. EFSA J, 9(6), 2207. https://doi.org/10.2903/j.efsa.2011.2207
- European Food Safety Authority. Scientific opinion on the substantiation of a health claim related to “native chicory inulin” and maintenance of normal defecation. 2015.EFSA J 13(1), 3951. https://doi.org/10.2903/j.efsa.2015.3951
- Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11(8):506-514. https://doi.org/10.1038/nrgastro.2014.66
- Guarner F, Schaafsma GJ. Probiotics. Int J Food Microbiol. 1998;39(3):237-238. http://doi.org/10.1016/S0168-1605(97)00136-0
- European Commission. Commission Implementing Regulation (EU) 2021/900 of 3 June 2021 authorizing the placing on the market of galacto oligosaccharides (GOS) as a Novel food. Official Journal of the European Union. 2021. https://eur-lex.europa.eu/eli/reg_impl/2021/900/oj/eng
- European Commission. Regulation (EC) No 1924/2006 on nutrition and health claims made on foods. Official Journal of the European Union. 2006. https://eur-lex.europa.eu/eli/reg/2006/1924/oj/eng
- Food and Agriculture Organization of the United Nations; World Health Organization. Guidelines for the evaluation of probiotics in food. London, ON: Joint FAO/WHO Working Group on Drafting Guidelines for the Evaluation of Probiotics in Food. International Scientific Association for Probiotics and Prebiotics (ISAPP).2002. https://isappscience. org › probiotic guidelines.
- Nagpal R, Kumar A, Kumar M, Behare PV, Jain S, Yadav H. Probiotics, their health benefits and applications for developing healthier foods: a review. FEMS Microbial Lett. 2012;334(1):1-15. https://doi.org/10.1111/j.1574-6968.2012.02593.x
- European Parliament and Council. Directive 2002/46/EC on the approximation of the laws of the Member States relating to food supplements. OJEU 2002. https://eur-lex.europa.eu/eli/dir/2002/46/oj/eng
- Global Prebiotic Association. China Regulatory Status.2025. https://prebioticassociation.org/prebiotic-regulatory-guide/china/
- Shimizu T. Newly established regulation in Japan: foods with health claim. Asia Pac J Clin Nutr. 2002;11(2): S94-96. https://doi.org/doi:10.1046/ j.1440-6047.2002.00007.x.
- Global Prebiotic Association. Japan Regulatory Status. 2025. https://prebioticassociation.org/prebiotic-regulatory-guide/japan/.
- Food Navigator-Asia. Japanese functional foods businesses legally bound to four new rules from Sep. 2024. Jul 8. https://www.foodnavigator-asia.com/Article/2024/07/08/japanese-functional-foods-businesses-legally-bound-to-four-new-rules-from-sep
- ISAPP 2025. https://isappscience.org/postbiotics-a-global-perspective-on-regulatory-progress/.
- Código Alimentario Argentino (CAA). Capítulo XVIII: Alimentos funcionales. Articulos 1383 y 1385. 2023. https://www.argentina.gob.ar/anmat/ codigoalimentario
- Administración Nacional de Medicamentos, Alimentos y Tecnología Médica (ANMAT). Disposición 4980/2005. Regulación de alimentos funcionales y suplementos dietarios.2005. https://www.argentina.gob.ar/normativa/nacional/disposici%C3%B3n-4980-2005-109568
- Agência Nacional de Vigilância Sanitária. ANVISA (2010). Resolução RDC N° 27/2010. https://es.scribd.com/document/760418307/RDC-27-2010
- Agência Nacional de Vigilância Sanitária (ANVISA) (1999). Resolução RDC nº 16, de 30 de abril de 1999. Dispõe sobre o regulamento técnico de procedimentos para registro dealimentos com alegações de propriedades funcionais e ou de saúde em sua rotulagem. Diário Oficial da União. 1999. https://www.gov.br/anvisa/pt-br/assuntos/fiscalizacao-e-monitoramento/propaganda/legislacao/legislacao
- Agência Nacional de Vigilância Sanitária (ANVISA). Resolução RDC nº 18, de 30 de abril de 2008. Aprova o regulamento técnico sobre probióticos e prebióticos. Diário Oficial da União. 2008. https://bvsms.saude.gov.br/bvs/saudelegis/anvisa/2008/rdc0018_24_03_2008.html
- Agência Nacional de Vigilância Sanitária (ANVISA). Instrução Normativa IN nº 60, De 23 de dezembro de 2016. Lista de alegações de propriedade funcional aprovadas. Diário Oficial da União. 2016. https://www.gov.br › inspecao › produtos-vegetal
- Ministerio de Salud. Reglamento Sanitario de los Alimentos (Decreto N° 977/96).2020. https://www.bcn.cl/leychile/navegar?idNorma=71271
- Ministerio de Salud. Decreto N° 309: Reglamento de suplementos alimentarios. 2015. https://www.bcn.cl/leychile/navegar?idNorma=194668
- Instituto de Salud Pública.Informe técnico sobre regulación de prebióticos en Chile.2021. https://www.ispch.cl › uploads› resoluciones
- Instituto de Salud Pública. Requisitos para declaraciones de propiedades saludables en alimentos.2022. https://www. bcn.cl/leychile/navegar?i=1105664&f=2018-01-27
- European Comission Codex Alimentarius, EFSA. 2025. https://food.ec.europa.eu/horizontal-topics/international-affairs/international-standards/codex-alimentarius_en
- Meteored. ¡Primer probiótico hecho en Chile sale al mercado! Y fue elaborado en startup liderada por mujeres.2025. https://www.meteored.cl/noticias/ciencia/primer-probiotico-chileno-sale-al-mercado-y-fue-elaborado-en-startup-liderada-por-mujeres.html
- Mercado Libre. Probióticos y Prebióticos. 2025. https://listado.mercadolibre.cl/probioticos-y-prebioticos
- Instituto Nacional de Vigilancia de Medicamentos y Alimentos (INVIMA). Resolución 333/2011. Alimentos con propiedades saludables. 2011. https://scj.gov.co › files › R_ MPS_0333_2011
- Instituto Nacional de Vigilancia de Medicamentos y Alimentos (INVIMA). Resolución 810/2021. Actualización de requisitos para declaraciones nutricionales. 2021. https://www.minsalud.gov.co › Resolución No. 81.
- Secretaría de Salud. México. Norm Oficial Mexicana NOM-086-SSA1-1994. Alimentos con propiedades funcionales. 1994. https://dof.gob.mx/nota_detalle.php?codigo=4890075&fecha=26/06/1996#gsc.tab=0
- Secretaría de Salud. México. Norma Oficial Mexicana NOM-218-SSA1-2011. Suplementos alimenticios. 2011. https://dof.gob.mx/normasOficiales/4643/salud/salud.htm
- Comisión Federal para la Protección contra Riesgos Sanitarios (COFEPRIS). Reglamento de Control Sanitario de Productos y Servicios. 2023. https://salud.gob.mx/unidades/cdi/nom/compi/rcsps.html
- Dirección General de Salud Ambiental (DIGESA) Perú. Decreto Supremo 007-98-SA. Reglamento sobre vigilancia sanitaria de alimentos. 1998. http://www.digesa.minsa.gob. pe › Codex › Ministerio de Salud (Perú). Resolución Ministerial 546-2013. Declaraciones de propiedades saludables en alimentos. 2013. https://www.gob.pe/institucion/minsa/normas-legales/239538-665-2013-
- Mishra B, Mishra AK, Mohanta Y.K. et al. Postbiotics: the new horizons of microbial functional bioactive compounds in food preservation and security. Food Prod Process Nutr 2024; 6: 28. https://doi.org/10.1186/s43014-023-00200-w.
- Grand View Research. Postbiotics Market Size, Share & Trends Analysis Report. 2024. https://www.grandviewresearch.com/industry-analysis/probiotics-market
- Lucintel. Postbiotic Market Report: Trends, Forecast and Competitive Analysis to 2030. 2025. https://www.lucintel.com/postbiotic-market.aspx
- Grand View Research. North America Digestive Health Supplement Market Size& Outlook. 2024. https://www.grandviewresearch.com/horizon/outlook/digestive-health-supplements-market/north-america
- European Food Safety Authority (EFSA). Postbiotic Safety and Efficacy Review. 2023. https://efsa.onlinelibrary.wiley.com/doi/10.2903/j.efsa.2023.7746
- Meticulous Research. Postbiotics Market Size, Share, Forecast and Trends Analysis by Source-Global Forecast to 2032. 2025. https://www.meticulousresearch.com/product/postbiotics-market-5380
- Kerry Group. Emerging Markets in Functional Ingredients. 2023. https://www.kerry.com kerry›pdf›investors
- Lallemand Health Solutions. Postbiotic Commercialization Strategies. 2024. https://www.lallemand-health-solutions.com/en/
- Morinaga Milk Industry. Clinical Applications of Postbiotics in Infant Nutrition. 2023. https://morinagamilk-ingredients.com/
- Szydłowska A, Sionek, B. Probiotics and Postbiotics as the Functional Food Components Affecting the Immune Response. Microorganisms 2023; 11(1): 104. https://doi.org/10.3390/microorganisms11010104
- U.S. FDA. Dietary Supplements on Foreign and Domestic Inspections. 2024. https://www.fda.gov/food/guidance-documents-regulatory-information-topic-food-and-dietary-supplements/dietary-supplement.
- Rodríguez B. El análisis documental de documentos digitales y/o multimedia. Revista Códice 2005; 1 (2): 19-20. https://core.ac.uk/download/pdf/290487308.pdf.
- Bowen GA. Naturalistic inquiry and the saturation concept: a research note. Qual Res 2008; 8(1): 137-152. https://doi.org/10.1177/1468794107085301.
- Bowen GA. Document Analysis as a Qualitative Research Method. Qual Res J 2009, (9)2: 27-40. https://doi.org/10.3316/QRJ0902027.
- Hagen-Zanker J, Mallett R. How to do a rigorous, evidence-focused literature review in international development: A Guidance Note. 2013. London: ODI. (www.alnap.org/help-library/how-to-do-a-rigorous-evidencefocused-literature-review-in-international-development).