Obesity is associated with a low grade inflammation which contributes to the development of insulin resistance and diabetes. The aim of this study was to assess the total saturated (SFAs), monounsaturated (MUFAs) and polyunsaturated fatty acids (PUFAs) in plasma from asymptomatic obese subjects and to determine the arachidonic/eicosapentanoic acid ratio [ARA/EPA] as a marker of inflammation, and its eventual association with ultrasensitive CRP. Fourteen obese (34.4 ± 11.1y.; BMI: 36.0 ± 4,5 kg/m2) and 12 normal-weight (30.6 ± 7.8y.; BMI: 23,6± 2,4 kg/m2) subjects were recruited and their plasma fatty acids were determined by gas chromatography. usCRP was higher in the obese subjects (p=0,01) and correlates with their body fat content. The percentages of SFAs, MUFAs, PUFAs were not affected in the obese subjects but their concentrations were increased, compared with the control group. However, no differences in the long chain PUFAs (DHA and EPA) concentrations or in the plasmatic ARA/EPA ratio were observed in these subjects. These observations do not support a relation between the ARA/EPA ratio and the presence of low grade inflammation evaluated by plasma usCRP in this group of asymptomatic obese subjects.
Key words: Obesity; inflammation, polyunsaturated fatty acids; arachidonic acid, eicosapentanoic acid; docosahexaenoic acid.
La obesidad está asociada con una inflamación de bajo grado que contribuye al desarrollo de la insulino-resistencia y de la diabetes. El objetivo de este estudio fue evaluar las concentraciones plasmáticas de ácidos grasos saturados (AGS), monoinsaturados (AGMI) y poliinsaturados (AGPI) en sujetos obesos asintomáticos y determinar el ratio ácido araquidónico/ácido eicosapentanoico [ARA/EPA] como un posible marcador de inflamación, con su eventual asociación con los niveles de proteína C reactiva ultrasensible (PCRus). Se reclutaron 14 sujetos obesos (34,4 ± 11.1 años; índice de masa corporal: 36,0 ± 4,5 kg/m2) y 12 normopeso (30,6 ± 7.8 años; índice de masa corporal: 23,6 ± 2,4 kg/m2); las concentraciones plasmáticas de ácidos grasos fueron determinados por cromatografía de gases. Los niveles de PCRus fueron más elevadas en los sujetos obesos (p=0,01) y correlacionaron con el contenido de grasa corporal. Los porcentajes relativos de AGS, AGMI, AGPI no se vieron afectados en los sujetos obesos, pero sus concentraciones plasmáticas se incrementaron en comparación con el grupo control. Sin embargo, no se observaron diferencias en las concentraciones de PUFAs de cadena larga (DHA, EPA y ARA) ni en el ratio ARA/EPA en los sujetos obesos. Estas observaciones no apoyan el uso del ratio ARA/EPA como un marcador de inflamación de bajo grado evaluada por PCRus en este grupo de sujetos obesos asintomáticos.
Palabras clave: Obesidad, inflamación, ácidos grasos poliinsaturados, ácido araquidónico, ácido eicosapentaenoico, ácido docosahexaenoico.
Laboratory of Microbiology and Probiotics and Laboratory of Energetic Metabolism, Institute of Nutrition and Food Technology (INTA), Univ. of Chile, Santiago, Chile.
Obesity is currently one of the main problems of public health in the world including developing countries (1). In Chile, its prevalence is currently of 7 % in children lower than 6 years of age, 17 % in school-age children and more than 25% in adults; this means that more than half of the Chilean population is overweight and about 3,4 millions persons are obese, resulting in a high burden for the society (1, 2). Obesity is associated with a low grade, silent, inflammation which contributes to the development of insulin resistance, type-II diabetes, metabolic syndrome and cardiovascular diseases (3). One of the factors related to this process is the increasing mass of the adipose tissue, characterized by the presence of hypertrophied adipocytes, the infiltration of activated macrophages and the release into the circulation of pro-inflammatory mediators that increase the expression of acute phase proteins such as C-reactive protein (CRP) (3, 4). Plasma ultrasensitive CRP (usCRP) is considered as a clinical marker of low grade inflammation, however it is not very selective as it can be raised in other clinical conditions. concentrations may increase due to other causes. Other factor implicated in the development of this inflammatory process is the plasma fatty acid (FA) composition which, in the short term, is influenced to some extent by the amount and quality of dietary fats (5-7). High proportions of long-chain polyunsaturated FAs (LC-PUFAs) and low proportions of saturated FAs (SFAs) in the diet have been associated with improved insulin function (6, 7). In addition, n-3-LC-PUFAs such as eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) are considered as potential anti-inflammatory agents due to their capacity to modulate cytokine release and the metabolism of adipose tissue and its inflammatory profile while n-6-LCPUFAs such as arachidonic acid (ARA) are considered as proinflammatory (8, 9). In consequence, dietary fat is qualitatively important in determining the equilibrium between n-6 and n-3 plasmatic FAs. Taking into account these observations, the ARA/EPA ratio has been proposed as a biomarker of inflammation (9); it has been recently observed, for example, that this ratio is increased in neurological disorders associated with silent inflammation such as autism and depression (10, 11). However, although an increase ARA/EPA ratio was observed during the development of inactivity-associated insulin resistance (12), data about its use in obese humans are scarce and controversial (13, 14). Taking into account the above considerations, we determined the plasma concentrations of fatty acids and their relative proportions in asymptomatic obese subjects and evaluate whether the eventual alteration of the ARA/EPA ratio corelates with the concentrations of usCRP.
The protocol was approved by the Ethics Committee of INTA and an informed consent was obtained from all subjects. Asymptomatic obese (BMI>30 kg/m2) and normal weight (18.5 - 24.9 kg/m2) volunteers of either sex, non-smokers and between 18 and 50 years of age were recruited. The study protocol was approved by the Ethics Committee for Research in Humans of INTA, University of Chile, in compliance with the Helsinki Declaration. Informed consent was obtained from all subjects before inclusion in the protocol. Asymptomatic obese (BMI>30) and normal weight volunteers of either sex, non-smokers and between 18 and 50 years of age were included in the study. Subjects with previous or currently associated chronic pathologies such as diabetes, nephropathies or liver cirrhosis were excluded from the study.
Anthropometric data were obtained from all the subjects and their whole body composition was determined by air displacement plethysmography (Bod-Pod, Body Composition System; Life Measurement Instruments, Concord, CA, USA), as previously described (10).
Blood samples were obtained in fasted individual to determine biochemical and lipid profiles as well as ultrasensitive C-reactive protein (usCRP). An aliquot of heparinized blood was centrifuged and sera were stored at -30ºC until processed to characterize qualiand quantitatively the total plasma fatty acids and to determine the ratio between the n-6 and n-3 LCPUFAs.
Total lipids from plasma were extracted and subsequently purified according to Bligh and Dyer (11); methyl ester derivatives of the total lipids were prepared according to Morrison and Smith, using tricosanoic acid (23:0) as internal standard (12). Fatty acid analysis was performed by gas chromatography using a Hewlett Packard 5890 series II Plus gas chromatograph equipped with a split injector and a flame ionization detector. Samples were injected on a 30-meter DB-FFAP capillary column (J&W, Folsom, CA, USA), using hydrogen as carrier gas. The column temperature was increased from 140 to 240ºC at 2ºC/min and the detector temperature was 260ºC.
The statistical analysis was carried out by using the Statistica vs. 7.0 software package (StatSoft, Tulsa, OK, USA). Results were expressed as means with standard deviation. The Shapiro Wilk test was used to determine the normal distribution of the different variables. As most of them displayed a skewed distribution, means between groups were compared by non parametric test (Mann Whitney U test) and Spearman’s rank test was used to determine correlations. Qualitative variables were compared by Chi-square. A p value <0.05 was considered as statistically significant.
12 controls and 14 obese volunteers were recruited in the study; their anthropometric and biochemical characteristics are shown in Table 1. As expected, most of the variables analyzed differed between the obese and control subjects; in particular, the usCRP level was significantly higher in the obese group compared with the controls (p=0.01) and a mild correlation was observed between their plasmatic usCRP and the percentage of body fat (r=0.52; p<0.05).
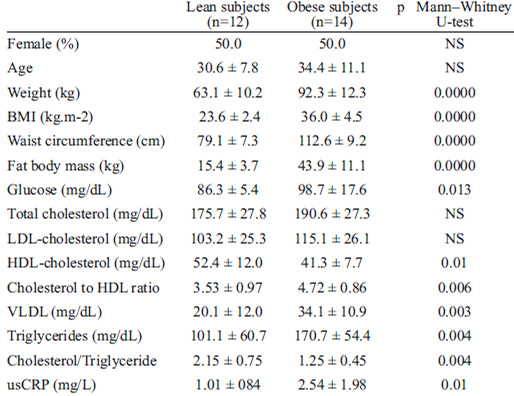
The plasma FA composition, expressed as percentage of total methyl-ester and concentrations (μg/ml), is shown in Table 2. Although the percentage of total SFAs did not change in the obese compared with the control subjects, their concentrations were increased by 39.6% (p=0.004), reflecting the respective increases of myristic (78.8%; p=0.0008), palmitic (43.5%; p=0.004) and stearic (25.9%) acids. Similarly, the percentage of total MUFAs was not altered in the obese subjects despite a slight increase in the percentage of palmitoleic (p=0.017); however, their plasmatic concentrations were significantly increased by 48.9% (p=0.002), due to the marked increase in palmitoleic (106.4%; p=0.002) and oleic (43.0%; p=0.004) acids. The percentage of total PUFAs did not differ in the obese compared with the control subjects, although the arachidonic and linoleic FAs percentages were slightly altered (p=0.004 and p=0.005, respectively). In these subjects, the plasma concentrations of total PUFAs were increased by 39.3% (p=0.002), reflecting the increase of linoleic acid (43.5%, p=0.0005), linolenic acid (104%; p=0.0003) and, in a minor proportion, of dihomo-γ-linolenic acid (34.9%), while ARA concentrations remained unchanged. Neither the percentage of EPA and DHA nor their plasmatic concentrations were affected in the obese subjects compared with the controls. As a consequence of these results, the n-6/n-3 PUFAs and the PUFAs/SFAs ratio were similar in both groups, as well as the ARA/EPA ratio. A moderate positive correlation was observed between usCRP levels and total SFA concentrations (r=0.48; p<0.05) ratio.
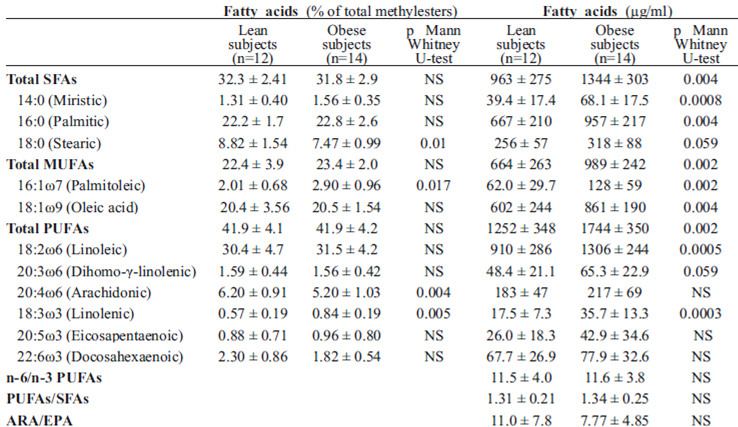
This study was carried out to evaluate whether the association between systemic low-grade inflammation and changes in FA composition and concentration in asymptomatic obese subjects. The presence of a low grade inflammation in these subjects was confirmed by the higher levels of usCRP, which positively correlated with their percentage of body fat mass, as observed in other studies (5-7).
On the other hand, plasmatic FAs, which reflects to some extent the amounts and composition of body stores and dietary fat (amount and quality of ingested fatty acids), seems to be linked to inflammatory activity in overweight individuals (5, 6). In this respect, a high LC-PUFAs/SFAs ratio has been associated with lower inflammation and improved insulin action. The n-6/n-3 LC-PUFAs ratio and more particularly the ARA/EPA ratio have also been proposed as markers of the inflammatory status. ARA is generally related to inflammatory processes due to its ability to modulate the production and release of pro-inflammatory eicosanoids and cytokines while EPA and DHA contributes to the endogenous formation of resolvins and protectins, molecules which are involved in the resolution of inflammatory processes (13). In consequence, the concentrations and plasmatic profiles of FAs may be important to determine the intensification or the improvement of the low-grade inflammation in the obese subjects. Our results show that there was virtually no difference among the different FAs when expressed as percentages of total methyl-esters, which implies that their proportion is conserved despite their higher amount in the obese group as compared to the normal-weight controls. However, when these results were expressed as plasmatic concentrations, important differences were observed between both groups. The increased amount of circulating SFAs, MUFAs and PUFAs in the obese group may be explained by the fact that these subjects have larger stores of FAs in adipose tissue and/or because their intake of dietary FAs is quantitatively different from that of normal-weight subjects (5, 14). It is important to note that neither the concentration of ARA nor those of EPA and DHA were affected in the obese group and that in consequence, the n-6/n-3 PUFAs and ARA/EPA ratios also remained unchanged. These findings are in opposition with those of other studies showing increases of ARA and decreases of EPA and DHA in obese subjects. For example, Fernandez-Real et al. have observed that the plasmatic concentrations of FAs in obese men and women remained unchanged while the proportion of some of these, particularly n-3-LC-PUFAs, were lower, compared to the lean subjects (7). It is possible that these discrepancies between studies were due to differences in the recruitment of the obese subjects or in the quality of their dietary fat.
In conclusion our results indicate that in young asymptomatic obese subjects, the total amount but not the proportion of fatty acids are likely to be related to low grade inflammation. In the subjects evaluated in this study, usCRP exhibits a moderate correlation with total SFAs and the LA/LNA ratio, but not wit the ARA/EPA ratio.
Recibido: 09-03-2011
Aceptado: 27-05-2011