Bee pollen has been reported to show antioxidant and radical scavenging activities; contributing to anti-inflammatory and gastroprotective properties. Venezuelan honeybee pollen has been little studied, but is consumed because its properties are known from other countries reports. On the basis of these reports, water, ethanol and methanol soluble fractions were prepared from dried bee-pollen commercially available and produced by La Montaña farm (Mérida, Venezuela). These fractions were evaluated for their functional properties, specifically, polyphenol content and total antioxidant activity. Pollen samples were separated by color in four fractions: yellow, brown, orange and ochre. Polyphenol content ranged between 396.7 to 1286.7 gallic acid equivalents GAE/100 g pollen; it was highest in pollen homogenates obtained with ethanol, followed by those obtained with methanol and water. The antioxidant activity ranged from 0.50 to 1.84 μmoles Trolox equivalents TEAC/100 g for water and ethanol homogenates respectively. The results presented in this work suggest that the ethanol extract of bee pollen show a potent antioxidant activity, comparable to human plasma, probably due to total polyphenol content of bee pollen. This is important because the bee pollen would be beneficial not only as a dietary supplement but also as a functional food.
Key words: Antioxidant activity, bee pollen, ethanol extract, Mérida, polyphenol content, Venezuela.
Se ha reportado que el polen de las abejas tiene actividad antioxidante y secuestra radicales libres; relacionada con sus propiedades antiinflamatorias y gastroprotectivas. El polen apícola de Venezuela ha sido poco estudiado, pero se consume gracias a las propiedades conocidas por reportes provenientes de otros países. Tomando como base estos reportes, se prepararon fracciones solubles en agua, etanol y metanol del polen apícola seco comercialmente disponible y producido por la Granja La Montaña (Mérida, Venezuela). Estas fracciones fueron evaluadas en cuanto a sus propiedades funcionales, específicamente, contenido de polifenoles y la actividad antioxidante total. Las muestras de polen fueron separadas en cuatro fracciones de acuerdo al color: amarillo, marrón, naranja y ocre. El contenido de polifenoles se encontraba entre 396,7 a 1286,7 equivalentes de ácido gálico EAG/100 g de polen, y fue mayor en los homogenatos obtenidos con etanol, seguido por aquellos obtenidos con metanol y agua. La actividad antioxidante varió entre 0.50 a 1.84 μmoles equivalentes de Trolox TEAC/100 g par los homogenatos de agua y etanol respectivamente. Los resultados presentados en este trabajo sugieren los extractos de etanol de polen de abejas presentan una potente actividad antioxidante, comparable al plasma humano, probablemente debida a su contenido total de polifenoles. Esto es importante ya que el polen de abejas podría servir no solo como un suplemento alimenticio sino como una alimento funcional.
Palabras clave: Actividad antioxidante, polen apícola, extracto etanólico, Mérida, contenido de polifenoles, Venezuela.
Departamento de Bioanálisis Clínico, Facultad de Farmacia y Bioanálisis, Departamento Ciencia de los Alimentos, Facultad de Farmacia y Bioanálisis, Departamento de Bioquímica, Facultad de Medicina, Universidad de Los Andes, Mérida 5101, Venezuela
Reactive oxygen species (ROS) are implicated in a wide range of human diseases including atherosclerosis, stroke, and diabetic retinopathy. When an imbalance between generated ROS and available antioxidants occurs, oxidative damage will spread via free radical generation in many cellular materials (e.g., DNA, lipids, and proteins). For this reason, the chemical natures and quantities of antioxidants in foods and medicinal plants have attracted much interest in recent years (1). Among them, honeybee-derived apicultural products, such as pollen, have been applied for centuries in alternative medicine as well as in food diets and supplementary nutrition due to their nutritional and physiological properties, mainly linked to the floral species or cultivars (2).
Bee pollen is a fine powder-like material produced by flowering plants pollen, mixed with nectar and bee secretions and gathered by the honey-bees. Pollens are the male reproductive cells of flowers and bees primary food source, containing concentrations of phytochemicals and nutrients and rich in secondary metabolites (3). It is known that bee pollen contains lipids, sugars, proteins, amino acids, vitamins, mineral substances, trace elements, carotenoids, polyphenolics such as flavonoids, and carbohydrates (4). Especially, the carbohydrates are derived from the nectar with which the flower pollen has been mixed in the flowers. The phenolic composition of pollen principally consists of flavonol glycosides and of hydroxycinnamic acids (5). This composition tends to be species-specific and has been related to the therapeutic properties (antibiotic, antineoplasic, antidiarrhoeic and antioxidant) of pollen (6). Flavonoids content are regarded as principal indicating ingredient substances of bee pollen and can be used for setting up quality standards in relation to their nutritional-physiological properties and for quality control of commercially distributed pollen preparations (7).
During ancient times, people throughout the world commonly used bee pollen, for the goodness and medicinal properties. In recent years, the physiological functionality of natural foods has received much attention, due to increasing interest in human health. Among natural products, honey bee derived apicultural products such as pollen and propolis have been applied for centuries in traditional medicine as well as in food diets and supplementary nutrition (8). In this sense, it has been reported that bee pollen prevents osteoporosis by increasing bone mass and exhibits antiallergic action (9-11). In addition, bee pollen has been reported to show antioxidant and radical scavenging activities (12-14), and recently, Akkol (15) have reported that antinociceptive (reducing sensitivity to painful stimuli), anti-inflammatory, gastroprotective and antioxidant effects of pure honey and honey-bee pollen mix formulation were evaluated comparatively.
On the basis of these reports, were prepared water, ethanol and methanol soluble fractions from dried honeybee-collected pollen commercially available and produced by La Montaña farm (Mérida, Venezuela). These fractions were evaluated for their functional properties, specifically, polyphenol content and total antioxidant activity (TAA).
Methanol [high performance liquid chromatography (HPLC) grade], 2,2´-azinobis(3-ethylbenzthiazoline-6-sulfonic acid (ABTS), sodium persulphate, and 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox) were purchased from Sigma Chemical Co. (St. Louis, MO). Ethanol, Folin-Ciocalteu reagent, sodium carbonate, and gallic acid were purchased from Merck (Darmstadt, Germany). Milli-QR plus water (Millipore, Bedford, MA) was used for all preparations.
Bee pollen samples were acquired from La Montaña farm (Mérida, Venezuela), where it is collected in pollen traps and sun-dried before packaging. Pollen samples were visually separated into four fractions according to the colors observed on white paper: 1 yellow pollen, 2 brown pollen, 3 orange pollen, and 4 ochre pollen. Each pollen fraction was ground in a mortar and was frozen until analysis. These colors were also observed in supernatants obtained with water, methanol and ethanol homogenization. Each biochemical test was performed by triplicate.
A weight of 0.10±0.01 g of each bee pollen fraction was placed on a glass homogenizer (Thomas No. A3528, USA), and 5 mL of Milli-QR plus water, methanol 99.9% (v/v) or ethanol 95% (v/v) were added, and homogenized on ice bath. Homogenates were centrifuged in a BHG Optima II (USA) centrifuge at 3,000 rpm by 10 min, and supernatants were used for biochemical analysis. Color was estimated by quantification of absorbance at 440 nm (Abs440) against a blank of water, methanol or ethanol, respectively.
Polyphenol contents were analyzed by spectrometry at 765 nm using Folin-Ciocalteu reagent (16). We mixed 100 μL of bee pollen extracts with 500 μL of Folin-Ciocalteu’s reagent diluted 1/10 with water, and added 400 μL of sodium carbonate 7.5% (w/v). The absorbance was recorded at 765 nm after 10 min of reaction at 37°C, against a blank with MQ water instead of sample. Polyphenol contents were measured with a calibration curve using a solution of 0.1 g/L of gallic acid as standard (0, 0.25, 0.05 and 0.1 g/L).
ABTS was dissolved in water to a 7 mM concentration. ABTS radical cation (ABTS+•) was produced by reacting 7 mM stock solution with potassium persulfate to a final concentration of 2,45 mM (in water), in the dark at room temperature (RT) for 12-16 h before use (17). For bee pollen samples analysis, ABTS+• solution was diluted with 20% ethanol (v/v) until 0.60-0.70 absorbance units at 735 nm and RT. Were taken 100 μL of bee pollen homogenates diluted in water, methanol or ethanol, and mixed with 7.5 ml of ABTS+• solution diluted in ethanol 20% (v/v). Were measured absorbance values 6 min after mixture. Was used a solution of 8 mM Trolox as antioxidant standard. Trolox was diluted to obtain 1, 2, 4 and 8 μM in 5 mM PBS buffer (pH 7.4). Was calculated decolorization percentage at 734 nm after 6 min and plotted as a function of different Trolox concentrations, and was reported TAA value of sample in comparison to the equation of the straight line obtained from this plot. TAA value for a given sample would be equivalent to Trolox concentration that produces the same decolorization percentage.
All experiments were done in triplicate. Data were analyzed by ANOVA, with average comparison through post hoc Scheffé test (18) using SPSS 12.0 (SPSS) software. Are indicated statistical differences to p< 0.05.
It is interesting to observe that each solvent (ethanol, methanol, water) produced a bee pollen homogenates of different colors, with biggest Abs440 values in homogenates using ethanol as solvent (Table 1).
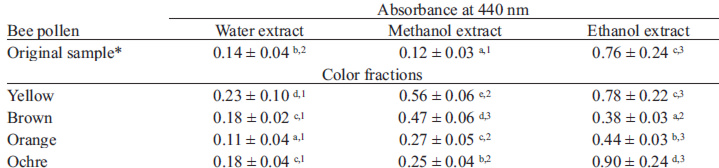
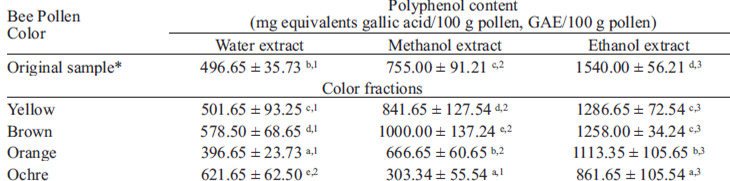
The highest polyphenol content was observed on bee pollen extracted with ethanol, followed by those obtained with methanol, and being lower in the water homogenates (Table 2). Besides, polyphenol content of ethanol homogenates is higher for yellow and brown pollen, and lower for orange and ochre pollen. In general lines, orange and ochre bee pollen present the lowest values of polyphenol content with every solvent used in this study (Table 2).
TAA values of pollen homogenates in different solvents are presented in Table 3. It can be seen that highest TAA is observed in bee pollen homogenized with ethanol, while water homogenates presented the lower TAA values. The highest TAA values are observed for ochre and brown pollen samples homogenized with ethanol.

In previous works with bee pollen from Cacute (Mérida, Venezuela), yellow pollen (Brassica napus, Brassicaceae), brown pollen (Zea mays, Poaceae), creamy pollen (Datura arborea, Solanaceae) and the orange pollen (Fraxinus americana Oleaceae) were identified by melissopalynology (19). Later, colors of bee pollen from Misintá another location in the Venezuelan Andes were identified as yellow (Brassica napus Brassicaceae, Rumex acetosella Polygonaceae), orange (Fraxinus americana Oleaceae, Guazuma ulmifolia Malvaceae, Hyptis suaveolens Labiatae), ochre (Coriandrum sativum Apiaceae, Eucalyptus sp. Myrtaceae, Zea mays Poaceae, Psidium sp. Myrtaceae), dark brown (Parkinsonia aculeata Fabaceae-Caesalpinioideae, Prunus persica Malvaceae) and green (Malus silvestris and Fragaria vesca Malvaceae) (20). More color classes were separated and other species identified in the brown (Medicago denticulata Fabaceae-Faboideae, Myrcia acuminata Myrtaceae and yeast), dark brown (Parkinsonia aculeata Fabaceae-Caesalpinioideae), and ochre (Cercidium praecox Fabaceae-Caesalpinioideae) fractions (21). Although in the present work melissopalynological analysis were not included, the above mentioned species are botanical references for bee pollen from the Venezuelan Andes.
In the present study was evaluated the TAA activity of bee pollen, previously separated in four color sets, homogenized with water, methanol and ethanol. In general, was observed that the descending order of antioxidative activities was ethanol preparations, followed by methanol homogenates, and last water extracts (Table 3). In the same way, the highest polyphenol content values were observed in ethanol bee pollen homogenates, followed by methanol homogenates and water homogenates (Table 2). Regarding to TAA values of different colors of pollen, the highest values of antioxidant activity were seen in brown and ochre bee pollen, especially in ethanol and methanol extracts. It is interesting to note that ethanol pollen extracts presented the highest absorbance 440 nm, TAA and polyphenol content values, (Tables 1, 2 and 3). Campos et al. (22) demonstrated that the flavonoid/phenolic components must play a significant role in the free radical capacity scavenging of bee pollen based on the observation that the bee pollen which exhibit the highest activity is that with the highest level of flavonoids and phenolic acid derivatives.
Maruyama et al. (2) studied anti-inflammatory effects of water and ethanol extract of bee pollen from Cistus sp. from Spanish origin. The pollen extracts were administered orally to rats, and one hour later paw edema was produced by injecting of 1% solution of carrageenan, and paw volume was measured before and after carrageenan injection up to 5 h. They found that the water extract showed almost no inhibitory activity of carrageenan-induced paw edema, but ethanol extract showed relatively strong inhibition of paw edema. Also, flavonoids were isolated and purified from the ethanol extract of bee pollen, and identified at least five flavonoids and their glycosides. Finally, the ethanol pollen extract presented a higher radical scavenging activity than water extract.
On the other hand, Graikou et al. (13) studied antioxidant activity of Greek pollen, but found that water extracts presented higher antioxidant activity than methanol extracts. Also, in water extract seven flavonoids have been isolated and identified by modern spectral means, and from the methanolic extract, sugars, lipid acids, phenolic acids and their esters have been also identified, which mainly participate to the biosynthetic pathway of pollen phenolics. In conclusion, the Greek pollen is rich in flavonoids and phenolic acids which indicate the observed free radical scavenging activity. In another research, Silva et al. (23) determined the free radical scavenging activities of different solvent extracts of pollen using DPPH assay. This activity varied according to the solvent used in each extract, and decreased in the order: EtOAc>EtOH>Hexane extract. It appears that the EtOAc extract of the pollen is a good scavenger of active oxygen species. As can be seen from these studies, the free radical scavenging activities and TAA activities depend of solvent used for extraction. Lotus (67.8 eq/g FRAP, 112.1 eq/g DPPH) and escalonia (69.5 eq/g FRAP, 119.9 eq/g DPPH) pollen from Chile showed that antioxidant activity was lower with FRAP than DPPH essays, and both results correlated negatively with Cu, Mn, Fe, Zn (24). The effective concentration (EC50) was used to measure the free radical scavenging activity with the DPPH assay to compare floral and bee pollen, which was over 28-fold) higher in the bee corn pollen (7.42 ± 0.12 μg/ml), than the floral corn pollen (212 ± 13.6% μg/ml) (25). These authors found 7-O-Rapigenin as the major active compound in bee pollen. The antioxidant activity of organic bee pollen from Portugal (Cistaceae Cistus, Boraginaceae Echium, Rosaceae Prunus, Fagaceae Castanea, Asteraceae Leontodon, Fabaceae Trifollium, Ericaceae Erica, Fagaceae Quercus, Mimosaceae Mimosa, Myrtaceae Eucalyptus, Rosaceae Rubus) varied between 3.0 ± 0.7 mg/mL with DPPH, and 4.6 mg/mL ± 0.9 mg/mL β-carotene bleaching assays values (EC50) (26).
Polyphenol content determined using the Folin-Ciocalteu reagent ranged from 396.7 to 1286.7 GAE/100 g pollen. This values are in concordance with those found by Freire et al. (27) in honey bee pollen collected from Bahia (Brazil), which are between 410.5 to 2130.2 mg GAE/100 g pollen; or by Morais et al. (14) who reported total phenolic content of five Portuguese honeybee pollen between 100.5 and 1600.8 mg GAE/100 g pollen. Both pollen samples from Brazil and Portugal above mentioned had high antioxidant activity. Pollen studied in this work showed a high antioxidant activity, similar and in some cases higher than plasma antioxidant system like ascorbic acid (1.05 ± 0.02 mM of Trolox equivalents), ±-tocopherol (0.97 ± 0.06 mM of Trolox equivalents), glutation (1.28 ± 0.04 mM of Trolox equivalents) y uric acid (1.05 ± 0.02 mM of Trolox equivalents) (17). A wide range of flavonoid types has been determined previously from pollen of different geographic origin among which mainly flavones, flavonols, dihydroflavonols and chalcones. Flavonoids are widely recognized as reliable chemotaxonomic markers not only of plants but also of bee pollen and honey; and the flavonoids in pollen and honey give special information of geographic and botanical origin of pollen (28).
To our knowledge, this is the first report on the biological activities of Venezuelan bee pollen. However, further studies are needed to clarify the type of flavonoids presented in Venezuelan honey bee pollen and possible mechanisms of action for flavonoids and other constituents of pollen. In conclusion, the results presented in this work suggest that the ethanol extract of bee pollen show a potent antioxidant activity, comparable to other found in plasma, probably due to the total polyphenol content of plant origin. This is important because the bee pollen would be beneficial not only as a dietary supplement but also as a functional food.
This work was supported by Consejo de Desarrollo Científico, Humanístico, Tecnológico y Artístico, Universidad de Los Andes (Mérida) CDCHTA-ULA Grant No. 1312-05-03-A.
Recibido: 20-07-2012
Aceptado: 22-12-2012