Aflatoxin M1 (AFM1) is found in milk and other excretion products after aflatoxin B1 intake. AFM1 is carcinogenic to humans, and known levels of dairy product contamination is important to understand the risks to which the population is exposed. The occurrence of AFM1 was evaluated in 42 milk samples commercialized in Londrina, Paraná State, Brazil and this rate of occurrence was used to estimate this exposure. AFM1 determina tion was ca rried out by ELISA, and was detected in 100 % samples at levels ranging from 0.01 to 0.81 μg/kg (mean 0.13 μg/kg). None of the samples p resente d AFM1 above the maximum permitted level by Brazilian Legislation (0.5 μg/kg for fluid milk and 5 μg/kg for milk powder). The estimated daily intake (EDI) of AFM1 was evaluated, and the average intake was 0.468 ng/kg body weight (b.w.) for adolescents, 0.384 ng/kg b.w. for adults and 0.559 ng/kg b.w. for the elderly. Values of EDI of AFM1 found in Londrina pose a toxicological risk to the population investigated. To the best of our knowledge, this is the first report on estimat ed AFM1 dietary exposure from Paraná, Brazil
Key words: Mycotoxins; aflatoxin M1 (AFM1); dairy products; ELISA; estimated daily intake
Aflatoxina M1 (AFM1) é encontrada no leite e em outros produtos de excreção após o consumo de aflatoxina B1. AFM1 é carcinogênica para humanos, e avaliar os níveis de contaminação em produtos lácteos é importante para conhecer os riscos aos quais a população está exposta. A ocorrência de AFM1 foi avaliada em 42 amostras de leite comercializadas em Londrina, Estado do Paraná, Brasil, e sua ocorrência foi utilizada para estimar sua exposição. A determinação de AFM1 foi avaliada por ELISA, e foi detectada em 100% das amostras, em níveis variando de 0,01 a 0,81 μg/kg (média 0,13 μg kg). Nenhuma das amostras apresentou níveis de AFM1 acima do máximo permitido pela Legislação brasileira (0,5 μg/kg para leite fluido e 5 μg/kg para leite em pó). A ingestão diária estimada (IDE) de AFM1 foi avaliada, e a ingestão média foi de 0,468 ng/kg de peso corporal (p.c.)/dia para adolescentes, 0,384 ng/kg p.c./dia para adultos e 0,559 ng/kg p.c./dia para idosos. Valores de IDE de AFM1 encontrados em Londrina supõem um risco toxicológico para a população investigada. Do melhor do nosso conhecimento, este é o primeiro trabalho sobre a exposição estimada de AFM1 do Paraná, Brasil.
Palavras chave: Micotoxinas; aflatoxina M1 (AFM1); produtos lácteos; ELISA; exposição diária estimada
Program on Milk Science and Technology, University North of Paraná (UNOPAR), Londrina, PR, Brazil.
Aflatoxins are fungus secondary metabolites that contaminate cereals and other products of vegetable origin. Aflatoxin B1 (AFB1) is the most common and the most toxic aflatoxin. AFB1 is amongst the most potent genotoxic and carcinogenic substances known to date and is classified as Group 1 by the International Agency for Research on Cancer – carcinogenic to humans (1).
After ingesting AFB1 contaminated feeds, a part is degraded in the rumen, resulting in the formation of aflatoxicol. The remaining fraction is absorbed in the digestive tract by passive diffusion and is hydroxylated in the liver to aflatoxin M1 (AFM1) (2). Circulating AFM1 can be excreted in the urine or appear in milk (3). This excretion is also observed in human milk. AFM1 was originally classified as Group 2B – possibly carcinogenic to humans, in 1993, but subsequent evidence of its cytotoxic, genotoxic and carcinogenic effects have led to a new categorization of AFM1 as Group 1 (1).
In Brazil, the national milk production in the first trimester 2014 was estimated at 6.2 billion liters (4), and Paraná State is the third major producer in the country. Since the presence of AFM1 and other contaminants in milk is of public health concern, the Ministry of Agriculture, Livestock and Food Supply instituted the National Plan for Residue Control (PNCR) in 1999 (5). The PNCR has the basic function to regulate, control and survey the food chain. Their actions aim to know and prevent the violation of secure levels or the maximum permitted levels of authorized substances, as well as the occurrence of residues prohibited in the country. The Milk PNCR stipulates the analysis of AFM1 in 100 milk samples per year by immunoassay ELISA or high performance liquid chromatography. Samples are collected randomly on a weekly basis by the Federal Inspection Service and sent to official or accredited laboratories.
The aim of the present study was to evaluate the natural occurrence of AFM1 in pasteurized, ultra heat treated milk and milk powder from Londrina, Paraná State, Brazil and estimate the daily AFM1 intake of consumers.
A total of 42 samples of pasteurized, ultra heat treated (UHT) milk and milk powder were collected randomly in July 2014 from supermarkets in Londrina, Paraná State, Brazil. Sample information was taken from the labels. Samples were produced in six different States of Brazil. According to information from the packaging, the pasteurized milk samples were produced in July 2014. The UHT milk samples were produced in March, April, May and June 2014. The powder milk samples were produced in December 2013, February, April, May and June 2014. Before being analyzed, the pasteurized milk samples were stored at 4 ºC and the UHT milk and milk powder samples at room temperature. The analyses were made within the expiration period.
Milk powder samples were weighed and homogenized with distilled water, warmed to 50 ºC in a water bath and prepared as fluid milk samples. The samples were defatted by centrifugation at 3500 g at 15 ºC for 10 min. The defatted supernatant was subjected to the ELISA test for AFM1 (Ridascreen, R-Biopharm AG, Germany). According to the manufacturer, the limit of detection is 5 ng/L for fluid milk (corresponding to 0.004 μg/kg), and 0.05 μg/kg for milk powder. Recovery rate in milk is 85 % and in milk powder, 96 %. The calibration curve was constructed with AFM1 standard solutions at 0, 5, 10, 20, 40 and 80 ng/L. A good correlation coefficient (r2) was obtained (0.98). Samples were analyzed in duplicate.
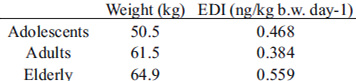
The intake estimates were based on the food consumption data collected by the National Household Food Budget Survey, POF 2008–2009, conducted by the Brazilian Institute for Geography and Statistics from May 2008 to May 2009. The data from the survey were presented as the average daily food intake (g/day) for three different age groups: adolescents (10 to 19 years old), adults (20 to 64 years old), and the elderly (over 65). The data referred to raw food consumption. The sampling survey consisted of 13,569 households and 34,003 inhabitants. The body weight (b.w.) was calculated as the arithmetic mean of the median from the age groups (6).
The estimates of dietary exposure to AFM1 were calculated from the amount of the compound found in dairy products (μg/kg), the daily intake of dairy products by the age groups (g), and the mean body weight of the age groups (Kg).
AFM1 levels in pasteurized, UHT milk and milk powder that did not fit normal distribution, were analyzed using the Kruskal-Wallis non-parametric test. Differences were considered to be significant at p < 0.05.
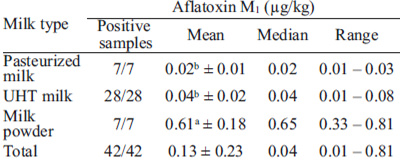
AFM1 was detected in 100 % samples (Table 1), with levels ranging from 0.01 to 0.81 μg/kg, and a mean of 0.13 μg/kg. Differences were observed in AFM1 levels in milk powder samples (0.61 μg/kg) compared to pasteurized (0.02 μg/kg) and UHT milk (0.04 μg/kg) (p < 0.05).
To calculate the estimated daily AFM1 intake through fluid milk and milk powder, the contamination of whole milk and skimmed milk was calculated (Table 2).
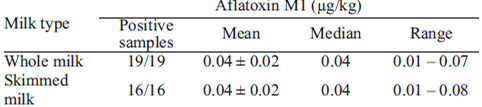
Determination of the exposure degree is one of the most important parameters to assess risk from chemical compounds. The Estimated Daily Intake (EDI) of AFM1 from fluid milk and milk powder (Table 3) was obtained using the amount of food consumed and the corresponding mean concentrations of AFM1 detected in each food group, taking into account the mean body weight of the age groups. Since all samples were positive for AFM1, the real concentration was used in the calculation (18). According to the IBGE (6), whole milk consumption was estimated at 38.6 g/day by adolescents, 31.5 g/day by adults and 45.6 g/day by the elderly. Skimmed milk consumption by adolescents, adults and the elderly was 2.5, 4.4 and 9.7 g/day, respectively. Milk powder consumption by adolescents, adults and the elderly was 0.3, 0.3 and 0.4 g/day, respectively. Considering the ingestion of these three dairy products, the AFM1 EDI ranged from 0.384 ng/kg b.w. per day for adults to 0.559 ng/kg b.w. per day for the elderly (Table 3).
AFM1 was detected in all samples investigated. It is well known that heat processing is not able to degrade mycotoxins (7, 8). However, in milk powder processing, the water elimination process produces the milk solids, and the heat-resistant contaminants such as mycotoxins concentrate.
Brazilian legislation sets a maximum permitted level of AFM1 for fluid milk of 0.5 μg/kg and for milk powder of 5 μg/kg (9). All the samples evaluated were within these values. The European Commission legislation (10) establishes a limit of 0.05 μg/kg of AFM1 in raw milk, heat-treated milk and milk for the manufacture of milk-based products. Two samples of pasteurized milk (28.6 %), five samples of UHT milk (71.4 %) and three samples of milk powder (42.8 %) exceeded this limit.
In Nigeria, AFM1 was detected in cow milk from 2.04 to 4.00 μg/L (11). In Jordan, Herzallah (12) observed very high levels of AFM1 in raw and pasteurized sheep, cow and goat milk, ranging from 0.16 to 5.23 μg/kg in samples collected in the winter. However, levels were lower than limit of detection of the method (0.05 μg/kg) in samples collected in the summer. The author attributed the difference to the low feed quality offered to the animals during the winter. In Morroco, AFM1 was detected in 13 samples (27%), ranging between 0.01 and 1 μg/kg (13). Four samples were above the European legislation limit.
Previous studies also detected AFM1 in samples from Brazil, but few studies were performed in Paraná State. Singular result of AFM1 in pasteurized milk was observed by Santos et al. (14), who did not find this contaminant in any of the 82 samples analyzed in a region of Paraná State. Oliveira et al. (15) analyzed 75 samples from Minas Gerais State and showed that 23 (30.7%) samples contained AFM1. Different from the present study, all of the contaminated samples were found to be higher than the maximum acceptable limits for fluid milk in Brazil. Samples of pasteurized and UHT milk from São Paulo (48 samples) were analyzed by Oliveira et al. (16). The authors observed a 77.1% frequency of positive samples, with levels of 0.06 μg/L for pasteurized milk and 0.07 μg/L for UHT milk. Also in São Paulo, Shundo et al. (17) investigated 125 samples, and a high AFM1 frequency was also observed (95.2%). Thirty three percent of the samples exceeded the limit established by the European Commission.
In the Lebanon, Hassan and Kassaify (19) reported a lower EDI of 0.14 ng/kg b.w. per day in local dairy products, such as fresh milk, yogurt and different types of cheese (akkawi, kashta, karishe, halloum and shanklish). Duarte et al. (20) observed an even lower AFM1 EDI through milk consumption of a Portuguese average adult citizen of 0.08 ng/kg b.w. per day. Similar values were reported by Cano-Sancho et al. (21) in Spain for the adult population of 0.305 ng/kg b.w. per day. Because aflatoxins are carcinogenic, international expert committees (22) did not specify a tolerable daily intake (TDI) for these substances and concluded that daily exposure as low as <1 ng/kg b.w. contributed to the risk of liver cancer. It was therefore recommended that levels should be reduced to as low as reasonably achievable. Kuiper-Goodman (23) has proposed a TDI for AFM1 determined by dividing the median toxic dose (TD50) by an uncertainty factor of 5000. The proposed value is 0.2 ng/kg of b.w., a value equivalent to a risk level of 1:100.000. Taking these values into account, the Londrina population is under toxicological risk to AFM1 exposure.
Levels of AFM1 in fluid milk and milk powder commercialized in Londrina, Paraná State, Brazil are below the maximum permitted levels established by Brazilian Legislation. However, considering the European Commission legislation, some samples exceeded the limit. The estimated daily intake of AFM1 through dairy products for adolescents, adults and the elderly in Londrina poses a toxicological risk for the population. The most effective method of controlling AFM1 in dairy products is to reduce the contamination of animal feed with AFB1 by applying preventive measures in terms of temperature and humidity control that reduce fungal growth.
The author thanks the Coordination for the Improvement of Higher Education Personnel – CAPES, for the fellowship of Post-Doctoral National Program granted to Joice Sifuentes dos Santos.
Recibido: 30-02-2015
Aceptado: 28-04-2015