 ,
Yunue Flores Ruelas1
,
Yunue Flores Ruelas1  ,
Salma Cortés Alvarez1
,
Salma Cortés Alvarez1  ,
Mario del Toro Equihua1
,
Mario del Toro Equihua1  ,
Karmina Sánchez Meza1
,
Karmina Sánchez Meza1  ,
Fátima López Alcaraz1
,
Fátima López Alcaraz1  ,
Carmen A Sánchez-Ramírez1
,
Carmen A Sánchez-Ramírez1 
Introduction. The obesity worldwide has produced an increase in obesity-related diseases and can be associated with low concentrations of 25-hydroxyvitamin-D. Also obesity and low physical activity can decrease sun exposure, so the aim was to correlate vitamin D intake with serum 25-hydroxyvitamin-D levels and to assess sun exposure habits in schoolchildren with obesity. Materials and methods. A correlational study was performed from January 2017 to January 2018 on 103 children between 6-12 years of age, with a body mass index ≥+2SD for age and sex, according to the World Health Organization. Blood samples were taken to determine the serum concentrations of 25-hydroxyvitamin-D, a nutritional survey to determine the vitamin D intake and a sun exposure questionnaire were applied. A Spearman correlation coefficient analysis was performed. Results. Forty-seven percent of the children were girls. The median years of age was 10. The median serum 25-hydroxyvitamin-D levels were 35.5 ng/mL, 74.8% had sufficient levels, 25.2% had insufficient levels. The median vitamin D intake was 214.7IU in boys and 231.9IU in girls. Regarding sun exposure, most of the children had excessive levels of sun exposure and inadequate sun protection practices. A positive correlation between vitamin D intake and serum 25-hydroxyvitamin-D was identified only in boys (rho=0.276, p=0.041). Conclusion. A positive correlation between vitamin D intake and serum 25-hydroxyvitamin-D levels was found in obese boys and excessive levels of sun exposure with inadequate sun protection practices in boys and girls. Arch Latinoam Nutr 2022; 72(2): 75-83.
Key words: 25-hydroxyvitamin D, obesity, schoolchildren, sun exposure, vitamin D.
Introducción: La obesidad se le ha asociado con distintas comorbilidades, bajas concentraciones séricas de 25-hidroxivitamina-D, sedentarismo que a su vez podría comprometer la exposición solar; por tanto, el objetivo fue relacionar la ingesta de vitamina D con los niveles séricos de 25-hidroxivitamina-D y determinar los hábitos de exposición solar en escolares con obesidad. Materiales y métodos. Estudio correlacional realizado de enero 2017 a enero 2018, en 103 niños entre 6 y 12 años, con un índice de masa corporal ≥+2DE para edad y sexo, según la Organización Mundial de la Salud. Se extrajo muestras sanguíneas para determinar las concentraciones séricas de 25-hidroxivitamina-D, se aplicó una encuesta nutricional para determinar la ingesta de vitamina D y un cuestionario de exposición solar. Se realizó un análisis del coeficiente de correlación de Spearman. Resultados. El 47% de los sujetos eran niñas. La mediana de edad fue de 10. La mediana de los niveles séricos de 25-hidroxivitamina-D fue de 35,5 ng/mL, el 74,8% tenía niveles suficientes, el 25,2% tenía niveles insuficientes. La mediana de la ingesta de vitamina D fue de 214,7UI en niños y de 231,9UI en niñas. Con respecto a la exposición solar, la mayoría de los niños presentaban una exposición excesiva y prácticas inadecuadas de protección solar. Se identificó una correlación positiva entre la ingesta de vitamina D y la 25-hidroxivitamina-D sérica en los niños (rho=0,276, p=0,041). Conclusión. Se identificó una correlación positiva entre la ingesta de vitamina D y los niveles séricos de 25-hidroxivitamina-D en niños obesos y exposición excesiva con prácticas inadecuadas de protección solar en niños y niñas. Arch Latinoam Nutr 2022; 72(2): 75-83.
Palabras clave: 25-hidroxivitamina-D, obesidad, escolares, exposición solar, vitamina D.
https://doi.org/10.37527/2022.72.2.001
Autor para la correspondencia: M.D., Ph.D. Carmen Alicia Sánchez-Ramírez, E-mail: [email protected]
Vitamin D deficiency and obesity are public health problems, whose prevalence continues to rise, making it necessary to clarify the causes of said increase. Those two pathologies have been shown to be closely related, given that the increase in adipose tissue is inversely related to serum 25(OH)D levels and higher parathyroid hormone (PTH) concentration, as a result of the large vitamin D storage capacity in adipose tissue, compromising the vitamin’s bioavailability. High concentrations of PTH enhance the entry of intracellular calcium into adipocytes, causing lipogenesis and predisposing to weight gain [1].
It is well-known that vitamin D is obtained through two routes: the intake of food containing vitamin D and sunlight. The latter is the main source. Ideally, sun exposure is recommended for vitamin D synthesis [2,3]. There has been an increase in studies conducted on pediatric populations that analyze the association of vitamin D with sun exposure. Some have identified a positive association, finding that a lack of sun exposure compromises serum 25(OH)D levels [4,5].
Studies have also reported alarmingly low vitamin D intake in Belgium, China, and Spain [6]. Vitamin D deficiency in schoolchildren and adolescents has been reported in Mexico, the United States, Chile, and Indonesia, indicating that a lack of physical activity and a diet low in vitamin D could be the cause of such hypovitaminosis. In the State of Colima, foods rich in vitamin D are readily available and there is a high rate of sunlight per day, leading to the assumption that there would be no vitamin D deficiency. However, there is a high prevalence of obesity in schoolchildren (17.5%) [7]. Therefore, the aims of the present study were to determine vitamin D intake and sun exposure habits and correlate them with serum 25(OH)D levels in obese schoolchildren in Colima, Mexico.
A cross-sectional study was conducted from January 2017 to January 2018 on 103 children from two public schools. They were selected through convenience sampling and invited to participate in the study. The inclusion criteria were: 6 to 12 years of age, a body mass index (BMI)≥+2SD for age and sex, according to the WHO [8], and participation acceptance from the parents or guardians. The non-inclusion criteria were the presence of dermatologic alterations, liver or kidney disease, intestinal malabsorption syndrome, treatment with antiepileptics or corticosteroids, and intake of calcium or vitamin D supplements. The variables measured were serum 25(OH) D levels, vitamin D intake, and sun exposure.
All the equipment used was calibrated and each protocol was completely standardized. Weight was measured with a TANITA digital scale (Ironman inner Scan BC-558/segmental) and height was measured using a portable stadiometer (Tanita HR-200, length 64-214 cm, 1 mm accuracy)[9].
Body mass index (BMI) was assessed using Quetelet’s equation. The Z-score of BMI/age was calculated with the WHO ANTHRO PLUS program and the cutoff point for obesity was ≥+2 SD[8].
Waist Circumference (WC) was measured using a fiberglass tape above the uppermost lateral border of the right ilium, at the end of a normal expiration, and was recorded to the nearest millimeter. WC was classified in percentiles according to the pattern in Mexican-American children published by Fernandez, et al [10].
A sample of venous blood (5 mL) through venipuncture was taken by a pediatric nurse from each of the participants, after an 8-10 hour fast, utilizing test tubes with no anticoagulant. The samples of venous blood were taken between January-July. Each sample was centrifuged at 1600 g for 10 min to obtain the blood serum, which was stored at -75°C. Serum 25(OH)D was measured by the Immundiagnostik ELISA assay (Bensheim, Germany) and the levels were classified as deficient, insufficient, or sufficient (<25 nmol/L, 25- 75 nmol/L, ≥75 nmol/L, respectively) [11].
The level of vitamin D intake was obtained by a trained nutritionist through the application of a food frequency questionnaire (FFQ) validated by the Instituto Nacional de Salud Pública (INSP), consisting of 112 reagents that estimate macronutrient and micronutrient consumption. The questionnaire was analyzed using the “Nutritional habits and nutrient consumption assessment system” software, which calculates the amount of nutrients consumed per day [7].
The questions were answered by the parents or guardians of each child. Each question had 10 response options, divided into five sections: 1) never, 2) less than once a month, 3) 1-3 times a month, 4) number of times per week, and 5) number of times per day. Only one option could be chosen and had to be as close to reality as possible.
The consumption frequency of vitamin D food sources, such as whole milk, fresh cheese, asadero cheese, fish, sardines, tuna, packaged cereal, and whole grain cereal was analyzed. We considered the recommended dietary intake (RDI) of vitamin D of 600IU/day and the estimated average requirement (EAR) of 400 IU/day [12]. The intake adequacy percentages were classified as deficient when consumption was <80% of the EAR (<320 IU/day) and sufficient when consumption was between 80-120% of the EAR (320 – 480 IU/day), according to the recommendations established by the Institute of Medicine (IOM) [13].
A questionnaire was applied to assess habits and protection behavior regarding the sun, skin type (phototype), and exposure time, evaluating the average number of hours per day (from 10:00 am to 4:00 pm), per week, and on weekends. That questionnaire is divided into 3 sections (for adults, minors 1-10 years of age, and adolescents (11-17 years of age). The children’s section contains 12 items and the adolescents’ section has 10 items, which were the ones applied [14]. [15]. It also includes a classification of the color of untanned skin, and we classified skin color into groups: group 1 very fair; group 2 fair; group 3 olive; group 4 light brown; group 5 dark brown; and group 6 very dark.
The Kolmogorov test was used to determine the data distribution, obtaining a non-parametric distribution (p=0.200). Medians, percentiles, frequencies and percentages were calculated. The correlation of serum 25(OH)D levels with vitamin D intake was determined through the Spearman’s correlation. The chi-square test was used to compare qualitative variables and the Mann-Whitney U test for the quantitative variables. All analyses were carried out with the Statistical Package for the Social Sciences (SPSS version 20), considering a p<0.05 as significant.
An informed consent was obtained from the persons exercising the legal parental rights of the minor. This study adheres to the guidelines of the 1975 Declaration of Helsinki and its later amendments. The project was approved by the Ethics and Research Committee of the Hospital Universitario Regional of the State of Colima, under registration number (2016/1/CR/CL/PD/113).
A total of 103 obese schoolchildren were included in the study. Forty-eight 48 (47 %) of the children were girls and 55 (53 %) were boys. The median years of age was 10 (9-11). There were no significant differences in the anthropometric measurements (weight, height) or the Z-score of the BMI/age, according to sex, as shown in Table I.
The median serum 25(OH)D levels in the study population was 35.5 ng/mL (27.4-40 ng/mL). When classifying the levels of 25(OH)D, 74.8 % (77) of the children had sufficient levels and 25.2 % (26) had insufficient levels. When comparing the levels according to sex, no significant difference was found (p=0.753). Because there were no schoolchildren with deficient or excessive serum 25(OH)D levels, the population was divided into two categories (sufficient and insufficient). The insufficient group consisted of 29 (28 %) children, with a median of 24.7 ng/mL (19-26.5 ng/mL) and the sufficient group consisted of 74 (72 %) children, with a median of 37.5 ng/mL (35-42.1 ng/mL). No significant differences in the anthropometric measurements (weight, height) or the Z-score of the BMI/age according to the group of sufficient and insufficient was identified, except for the waist circumference being statistically significant higher in the group of insufficiency (0.029) (table I).
According to the data provided by the FFQ, vitamin D intake was below the EAR, at 224.88 IU (37.48 % of the RDI). The mean vitamin D intake was 232.2 ± 121.8 IU in boys and 216.5± 101.4 IU in girls. When comparing vitamin D intake according to sex, no significant difference was found (p=0.523). The adequacy percentages were determined, showing that 84.5% (45 girls and 42 boys) had deficient vitamin D intake and 15.5% (3 girls and 13 boys) had sufficient intake. When compared by sex, vitamin D intake was deficient more frequently in girls, with statistical significance (p= 0.014).
Regarding the frequency of consumption of vitamin D food sources, 1 glass of milk (2-3 times a day) was the most recorded, followed by cheese and packaged cereal (2-4 times per week), and fish, including tuna(1-3 times per month). Foods never consumed were sardines (84.5%), cereals with fiber (82%), and beef/chicken liver (79.6%).
When comparing the frequencies of vitamin D dairy product consumption, according to sex, cheese was consumed more frequently only by boys (p=0.048). Regarding the rest of the foods, there were no significant differences between the sexes. When comparing vitamin D intake between IU (113-288.2 IU), with a tendency toward lower intake in the insufficient group, but with no significant differences (p=0.196). There were also no significant differences in the consumption of dairy and animal sources of vitamin D between the two groups. With respect to cereal consumption, a statistically significant difference (p=0.034) was found between the sufficient and insufficient groups, with the former consuming more packaged cereal.
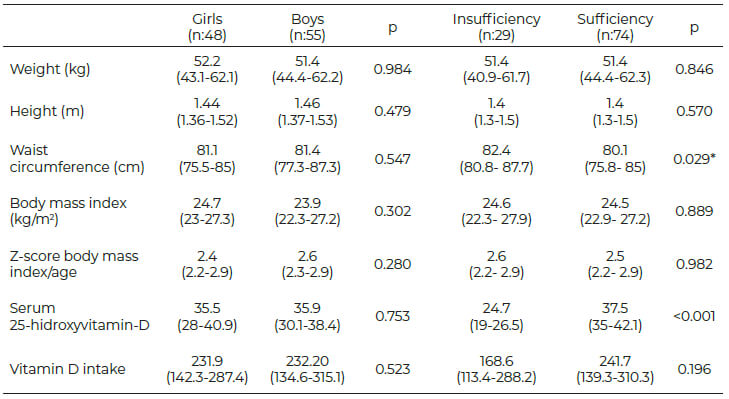
The correlation between serum 25(OH)D levels and vitamin D intake was not significant (rho=0.055, p=0.581) in the entire sample. However, the correlation between serum 25(OH)D levels and vitamin D intake, according to sex, was significantly positive for boys (rho=0.276, p=0.041) (Figure 1).
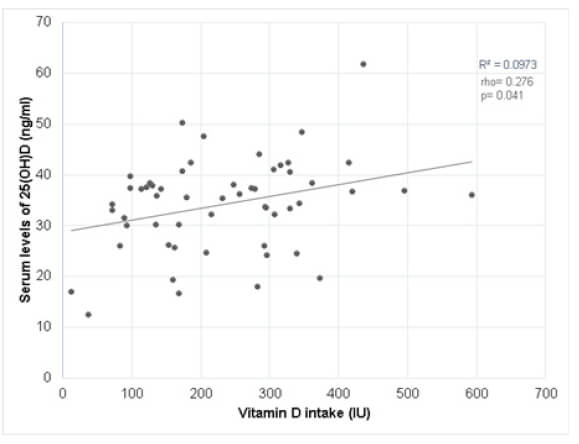
Regarding sun exposure, 49.5 % (51 children) had high sun exposure and 50.5 % (52 children) had moderate sun exposure. The table 2 shows the descriptive results of the attitudes according to sex, finding that boys wore a hat or cap more frequently than girls (p=0.043). There were no differences between boys and girls in relation to the other attitudes. On the other hand, the highest percentage of children sometimes or rarely used sunscreen, with only one-quarter of them sometimes using it. Sixty-eight percent of the total population always wore short-sleeved shirts and 64% never wore a hat, cap, or dark glasses, which are attitudes that favor vitamin D synthesis. With respect to staying in the sun or the shade, 37 % often or always sought to be in the shade.
When evaluating sun exposure time in minutes or hours per days of the week, 30% (31 children) were exposed >31 minutes, followed by 2 hours in 24.3 % (25 children). On the weekend, 28.2 % (29 children) were exposed >31 minutes and 25.2 % (26 children) for 2 hours. The correlation between the serum 25(OH)D levels and the sun exposure was no significant (rho=0.079, p=0.429).
According to untanned skin color, 48.5 % (50) had light brown skin, 24.3 % (25) olive, 14.6 % (15) fair, 5.8 % (6) very fair, and 1 % (1) very dark. Untanned skin color was classified into two groups (group A=groups 1-3; group B= groups 4-6) with no differences between boys and girls. When comparing the two skin color groups, according to the sufficient and insufficient groups, group B more frequently presented with insufficient/deficient vitamin D intake than group A. When stratified by sex, the girls with group 4-6 skin color more frequently presented with insufficient/deficient vitamin D intake (84.6 %) than the boys with group 4-6 skin color (62.5 %). Said difference was significant in girls and not in boys (p=0.036 and p=0.211, respectively).
High frequencies of obesity and hypovitaminosis D have been reported worldwide. We sought herein to correlate vitamin D intake and sun exposure with serum vitamin D levels, highlighting important considerations. Only obese children were included in the present study. Almost one-third presented with insufficient vitamin D intake and none presented with deficient or excessive intake. Said values are similar to those reported in a study conducted in Northwestern Mexico, in which 36% of the children with overweight or obesity had low 25(OH)D levels [16]. A study conducted in India reported deficient vitamin D intake in 94.7% of the obese children and adolescents analyzed [17]. In a Chilean study, the authors reported that 49% of obese children 9.6 ± 0.5 years of age had vitamin D deficiency [18], whereas authors in Canada found that 76 % of obese children and adolescents between 2-18 years of age had a suboptimal vitamin D status [19]. In a study conducted in Cuernavaca, Mexico, on 533 children and adolescents between 5 and 20 years of age, serum 25(OH)D levels were below 30 ng/mL, considered insufficient, in 90% of the study population[20]. Regarding anthropometric measurements, we identified a higher WC in the insufficient group of vitamin D, similar to what has been reported by our group and other authors [21]. Another study performed in pediatric population in Iran found similar results when analyzing the association between vitamin D status and different anthropometric measures, in which vitamin D deficiency was associated with WC and BMI [22].
It is important to highlight that no child in the present study met the vitamin D requirements issued by the IOM (600 IU), revealing that the children evaluated consumed half of the EAR (400 IU) for non-overweight and non-obese schoolchildren, which can affect serum 25(OH)D levels. That phenomenon has been identified in several countries that report alarmingly low vitamin D intake values [6]. In Belgium, 22% of children had inadequate vitamin D intake, and in China, vitamin D intake was below 10 μg/day in 86.1% of the school population and was associated with a low frequency of vitamin D intake from food [23].
A positive and significant correlation between vitamin D consumption and serum 25(OH)D levels was found in the boys analyzed, which could be explained by their having a higher consumption of foods rich in vitamin D than the girls. Organizations such as the National Institutes of Health (NIH) have issued special recommendations for obese children, such as frequent consumption of fortified milk, fish, fish oil, milk derivates, and fortified cereals to ensure adequate serum 25(OH)D levels, given that the requirements to cover the RDI must be 2 to 3-times higher than the RDI for non-obese children (600-1000 IU) [24]. In the present study, low consumption of vitamin D-rich foods, such as sardines, fortified cereals, beef or chicken liver, and fish was found, which could explain why 100% of the RDI was not met. That is similar to the results of a study conducted in the Czech Republic, in which low vitamin D intake (2.5-4.9 μg/day) was found and its main sources were eggs, pastries, milk and dairy products, meat, fish, and margarines [25]. In Spain, vitamin D food sources, such as fish, eggs, dairy products, and fortified cereals, were identified as contributing 85% of the total vitamin D intake, without being able to cover the recommended RDI [26]. In Asian countries, deficient consumption of vitamin D food sources, such as fish, fish viscera, or fish oil, has been reported [27].
When analyzing the consumption of vitamin D food sources, according to the sufficient and insufficient 25(OH)D groups, the schoolchildren that had sufficient intake had higher consumption of fortified cereal than those in the insufficient group. That could be explained by the fact that in Mexico, the Official Mexican Standard NOM-086-SSA1-1994 modifies food composition by fortifying and enriching foods [28]. With respect to vitamin D, the fortified products cover 25 % of the RDI.
In relation to sun exposure, the recommendation (depending on day, season, latitude, and skin pigmentation) is to expose arms and legs to sunlight at intervals of 5 to 30 minutes between 10 am and 3 pm twice a week to maintain adequate serum vitamin D levels [1]. According to the questionnaire applied, most of the children included in the present study had excessive levels of sun exposure and inadequate sun protection practices, which should promote vitamin D synthesis. That can be explained by the high ultraviolet index in the State of Colima, as well as its latitude, which favors prolonged sun exposure. In addition, attitudes identified in relation to sun exposure favored vitamin D synthesis in both sexes. However, despite favorable sun exposure for vitamin D synthesis, education about effective sun protection habits must be performed to avoid prolonged sun exposures, since it takes 20 minutes’ day to maintain and adequate level of vitamin D levels. A recently published study conducted in Spain in university students found similar results of those of our study, given that the students were potentially exposed to UV radiation without adopting adequate sun protection habits [29]. Another study performed in San Paolo, Brazil, on 155 healthy children, aged 6 to 24 months, found no association between the serum 25(OH)D levels and the sun exposure [3], whereas in China, 16,755 children (0-6 years of age) were evaluated, and the lack of sun exposure was found to compromise serum 25(OH)D levels [5].
Regarding untanned skin color, children in groups 4-6 presented with vitamin D insufficiency more frequently, with a significant difference in girls, whereas a study conducted by Al-Daghri et al. reported that dark-skinned adolescent boys had significantly lower serum concentrations of 25(OH)D, with limited sun exposure [30]. That could be explained by the reverse correlation between cutaneous pigmentary system activity (melanin production) and vitamin D production described by most of the experts in the field of vitamin D research [31].
Among the limitations of the present study is the fact that convenience sampling, rather than randomized sampling, was carried out, preventing the sample from being representative and the stages of Tanner were not performed. One of the study’s strengths was the use of a dietary survey validated for the Mexican population and a validated survey on attitudes regarding sun exposure designed for the age group evaluated.
In conclusion, the present study is the first in Mexico to verify the relationship of vitamin D intake with serum 25-hydroxyvitamin-D concentrations and to assess the sun exposure habits in schoolchildren with obesity, finding a positive correlation between vitamin D intake and serum 25-hydroxyvitamin-D levels in boys and excessive levels of sun exposure with inadequate sun protection practices in boys and girls.
The authors wish to thank Gusti Gould de Pineda for the English language revision of the present manuscript.
The authors declare that they have no conflict of interest.
Recibido: 23/08/2021
Aceptado: 26/04/2022